CAR T cell therapy has dramatically transformed the landscape of treatment options for numerous blood-based malignancies, yet it has demonstrated limited effectiveness when confronting solid tumors, which represent more than 85% of all cancer cases. Researchers at Columbia University have now developed an innovative approach using HIT cells—a highly sensitive variant related to CAR T cells—that successfully addresses a primary barrier in treating solid tumors with cellular therapies. In preclinical studies, this therapy completely eradicated kidney, pancreatic, and ovarian cancers in mouse models. The groundbreaking findings are detailed in the prestigious journal Science.
This pioneering research was carried out by a team from the Columbia Initiative in Cell Engineering and Therapy (CICET). Michel Sadelain, who serves as the director of CICET, is renowned for his foundational work in developing the CAR T therapies currently in use, which involve reprogramming a patient’s own immune cells into precise killers capable of hunting down and eliminating cancerous cells. Over recent years, his laboratory has been at the forefront of advancing HIT cell therapy as a next-generation solution.
“Tackling solid tumors remains a formidable challenge, but our latest advancements address a crucial element of this complex puzzle,” states Sadelain.
The Challenge of Solid Cancers
While multiple factors are known to hinder the performance of CAR T cells in solid malignancies, the initial and most fundamental hurdle for any cellular therapy is the ability to identify and target every single cancer cell within the tumor microenvironment. In blood cancers, malignant cells are typically straightforward targets for CAR T therapies because they are densely populated with CD19 molecules, which function as clear navigational signals guiding the engineered cells directly to the threat.
In contrast, solid tumors exhibit far greater diversity among their cellular components, lacking a universal molecular marker that could serve as the exclusive focus for therapeutic interventions. “Various molecules have been pinpointed that appear on 25%, 50%, or even 75% of tumor cells,” explains Sadelain, who also holds the position of Herbert and Florence Irving Professor of Medicine at Columbia University Vagelos College of Physicians and Surgeons.
“A treatment strategy aimed at these markers might succeed in wiping out a significant portion of the tumor mass, but achieving a complete cure requires the elimination of virtually every last cancer cell—anything less leaves the door open for recurrence.”
CD70 as a Key Marker in Many Solid Cancers
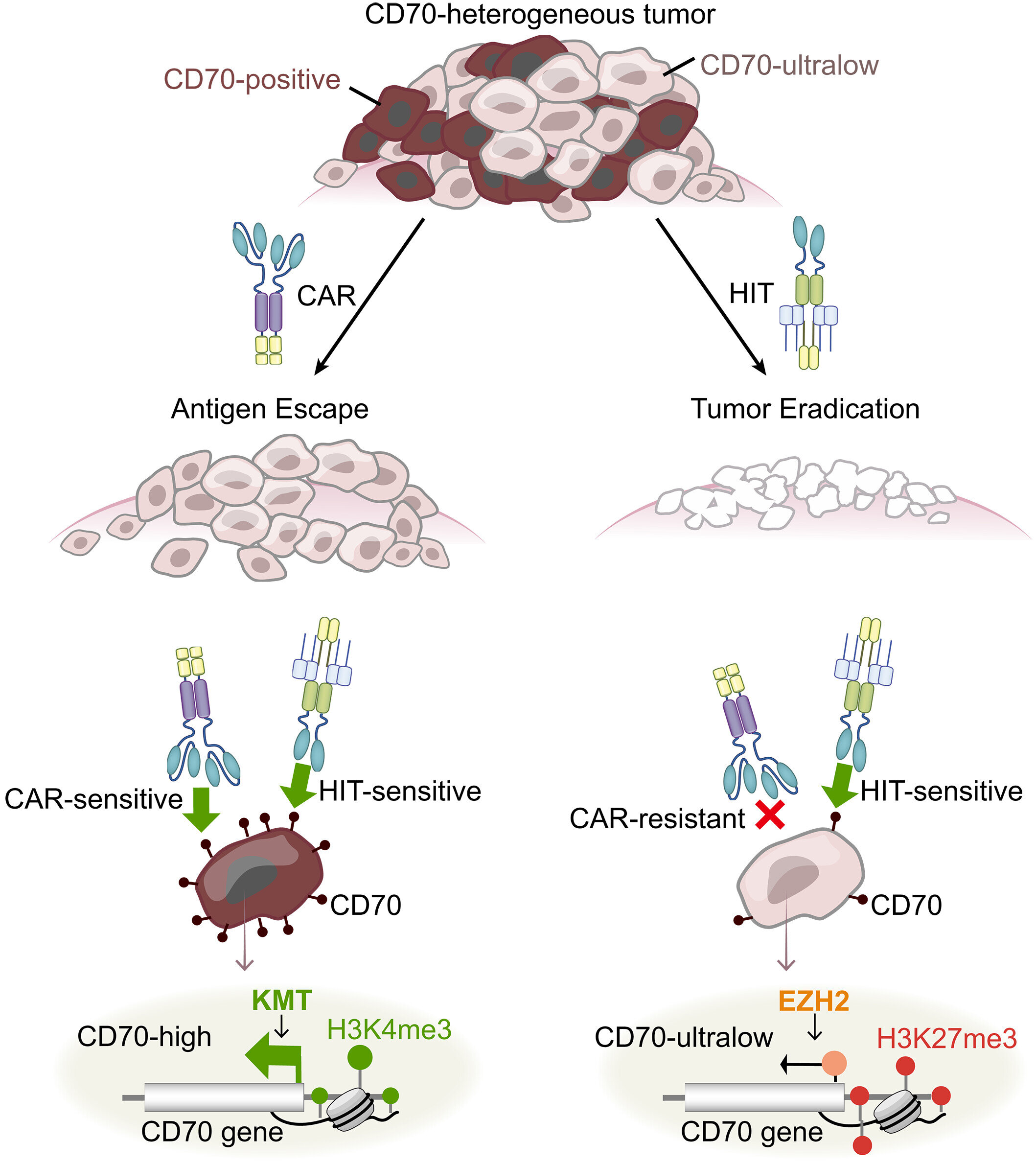
The latest investigation reveals an unexpected breakthrough: for certain categories of solid tumors, the protein CD70 emerges as a viable homing signal suitable for directing cell-based therapies. Although prior research indicated that CD70 expression fluctuated inconsistently across individual cells within a tumor, Sophie Hanina, the lead author and a research associate scientist at CICET, suspected that earlier detection techniques had overlooked cells bearing minimal quantities of this molecule.
By devising novel, ultra-sensitive detection protocols, Hanina confirmed that while CD70 levels can differ substantially from one cancer cell to another, every cell in the examined tumors displayed at least trace amounts of the protein on their surface membranes.
Solid Cancers Demand the Precision of HIT Therapy
To harness this discovery for practical therapeutic application, Hanina collaborated on the HIT platform, an emerging cell therapy technology being refined in Sadelain’s CICET laboratory. Standard CAR T cells are equipped to recognize only those cancer cells expressing abundant quantities of the designated target antigen. Hanina’s experiments demonstrated that CAR T cells engineered against CD70 struggled significantly against solid tumors in lab settings—a key reason why such therapies have underperformed in clinical scenarios involving solid malignancies.
“HIT cells represent the evolutionary successor to traditional CAR T cells,” Hanina elaborates. “They retain the programmable nature of CAR T constructs but possess the exquisite sensitivity of native T cells, enabling them to detect and respond to cancer cells displaying merely trace levels of target molecules.”
In rigorous mouse model experiments, HIT cells specifically programmed to target CD70 achieved total tumor regression in cases of pancreatic, kidney, and ovarian cancers. By comparison, conventional CAR T cells managed only partial tumor clearance. Moreover, the HIT cells exhibited remarkable specificity, sparing healthy tissues in the animals, as CD70 is predominantly absent from non-cancerous cells throughout the body.
Gearing Up for Human Clinical Trials
Building on these compelling preclinical results, Hanina and Sadelain are actively preparing to evaluate CD70-targeted HIT cells in human patients suffering from ovarian cancer and other relevant malignancies at Columbia University Irving Medical Center.
This promising therapy holds potential applicability across approximately 20 additional cancer types, such as glioblastoma and pancreatic adenocarcinoma, where CD70 expression has been documented to varying extents.
Despite the array of additional challenges posed by solid tumors to cell therapies—like immunosuppressive microenvironments and physical barriers—for those cancers that express CD70, ensuring comprehensive detection of all malignant cells stands out as a pivotal step toward success.
“Emerging evidence points to the evasion of low-antigen cancer cells as the primary reason for suboptimal outcomes with standard CAR T approaches,” Hanina notes. “We are optimistic that our CD70-specific HIT cells will provide the means to fully eradicate tumors by leaving no cancerous cell undetected.”