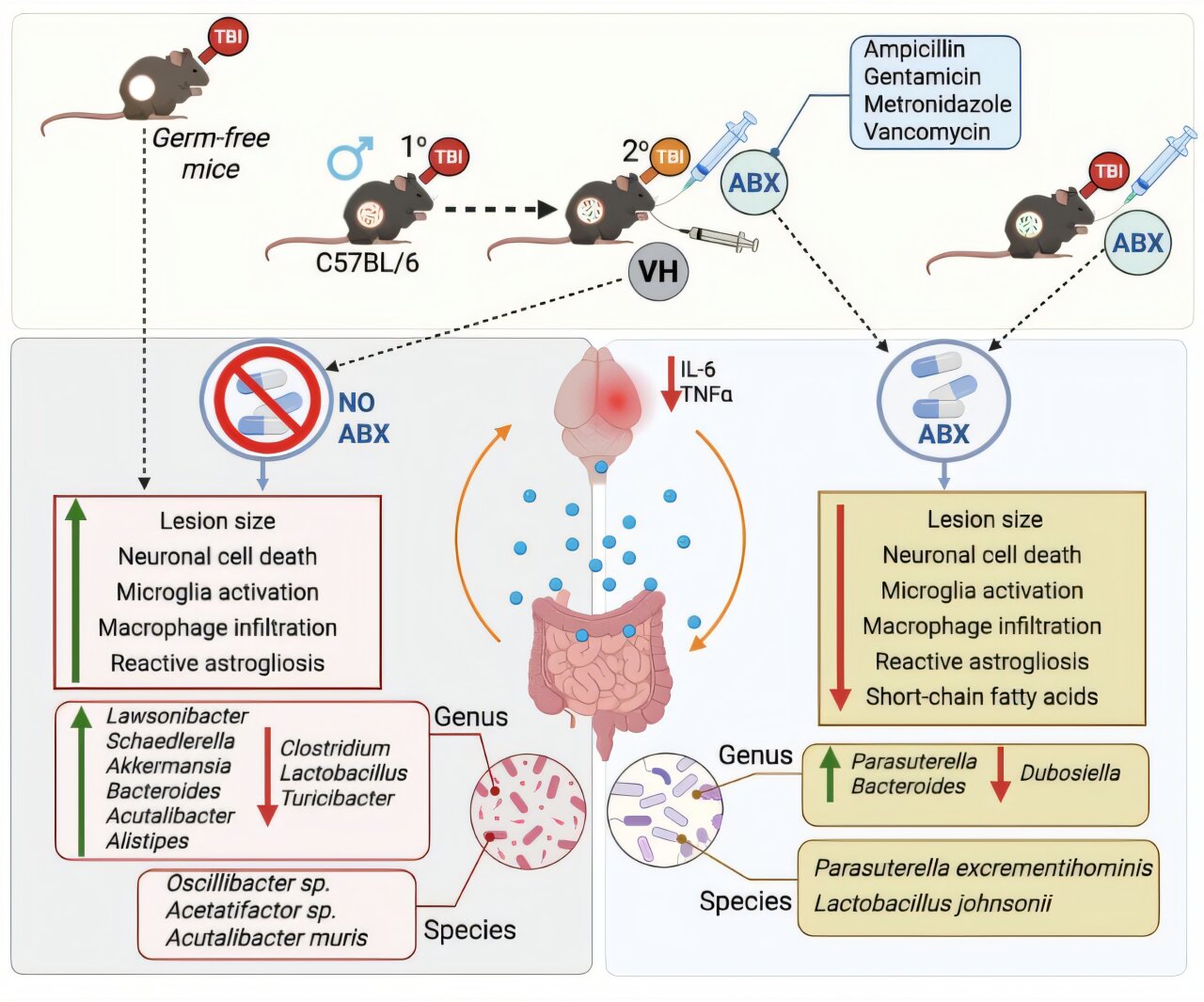
Researchers from Houston Methodist, under the leadership of Sonia Villapol, Ph.D., have published groundbreaking findings in Communications Biology, demonstrating that a brief course of antibiotics can substantially diminish neuroinflammation and neurodegeneration in the aftermath of traumatic brain injury (TBI). This effect is achieved through targeted modifications to the gut microbiome in experimental animal models.
Villapol, who serves as an associate professor within the Department of Neurosurgery at Houston Methodist, elaborated on the key discoveries: “Our investigations revealed that administering antibiotics after a TBI event effectively eliminates detrimental gut bacteria, shrinks the size of brain lesions, and minimizes neuronal cell death.” She further emphasized the pivotal role of the gut-brain connection, explaining, “These outcomes underscore a crucial gut-brain pathway where shifts in the microbiome composition impact peripheral immune responses, which subsequently influence neuroinflammatory processes following TBI.”
The mechanism behind this therapeutic approach involves the use of antibiotics to purge harmful bacterial populations from the gut, thereby creating an environment conducive to the proliferation of beneficial microbes. The research specifically pinpointed two beneficial bacterial strains—Parasutterella excrementihominis and Lactobacillus johnsonii—as essential contributors to cellular repair mechanisms. Villapol noted that these bacteria not only facilitate tissue regeneration but also play a significant role in modulating systemic inflammation throughout the body.
It is noteworthy that approximately 70% of the body’s immune system regulation originates from the gut microbiome. Disruptions in this microbial equilibrium, known as dysbiosis, can trigger widespread physiological disturbances due to the bidirectional communication along the brain-gut axis. This interconnected signaling network allows for mutual influence between the central nervous system and the gastrointestinal tract.
Villapol provided additional insight into the dynamics at play: “The brain continuously transmits regulatory signals to various organs across the body. In the wake of a traumatic brain incident, these signals can become distorted, leading to dysfunction in distant systems such as the digestive tract.” She cautioned that prolonged gut dysbiosis could impede the brain’s innate recovery processes, exacerbating long-term damage.
In the United States alone, healthcare providers encounter roughly 4 million cases of traumatic brain injuries annually. Emerging evidence suggests that the gut microbiome dysbiosis triggered by TBI might contribute to the onset of progressive neurodegenerative conditions, including Parkinson’s disease, Alzheimer’s disease, and various forms of dementia. This connection highlights the potential for microbiome-targeted interventions to alter disease trajectories.
The laboratory led by Villapol is dedicated to exploring innovative neuroprotective strategies aimed at combating inflammation associated with neurodegenerative pathologies. “By interrupting the cycle of neuroinflammation during either the acute or chronic phases post-injury, we may substantially lower the likelihood of individuals developing Alzheimer’s disease or dementia,” Villapol affirmed.
Looking ahead, the research team plans to advance their work by bioengineering the promising bacteria Parasutterella excrementihominis and Lactobacillus johnsonii. This next-generation approach seeks to engineer precision therapies capable of more effectively suppressing neuroinflammation and promoting neural repair.
The collaborative effort included contributions from several key team members: Hannah Flinn, Austin Marshall, Morgan Holcomb, Marissa Burke, Goknur Kara, Leonardo Cruz-Pineda, Sirena Soriano, and Todd J. Treangen. Their combined expertise was instrumental in unraveling the complex interplay between gut microbiota and brain health recovery.
This study opens new avenues for therapeutic development in TBI management. Traditionally, treatments have focused primarily on the brain itself, but these findings advocate for a holistic perspective that incorporates gut health as a modifiable factor in neurological recovery. The implications extend beyond acute injury response to long-term prevention of secondary neurodegenerative complications.
Understanding the gut-brain axis in greater depth could revolutionize clinical protocols for TBI patients. For instance, routine microbiome assessments post-injury might guide personalized antibiotic regimens or probiotic supplements to optimize healing outcomes. Moreover, this research challenges conventional wisdom by positioning short-term antibiotic use—not typically associated with brain health—as a potential ally in neuroprotection.
The bidirectional nature of the gut-brain axis means that interventions targeting the gut can yield measurable benefits in the brain. Beneficial bacteria like those identified in the study produce metabolites and signaling molecules that cross into systemic circulation, influencing immune cell activity and reducing pro-inflammatory cytokines that exacerbate brain damage.
Neuroinflammation, a hallmark of TBI pathology, involves the activation of microglia and astrocytes, leading to the release of damaging factors. By restoring gut microbial balance, antibiotics indirectly temper this inflammatory cascade, preserving neural tissue integrity and function.
Future clinical translations will need to address challenges such as antibiotic resistance and the need for human-specific microbiome profiles. Nonetheless, the preclinical success in animal models provides a strong foundation for translational studies, potentially leading to adjuvant therapies that complement existing standards of care like surgical intervention and rehabilitation.
Publication details: Hannah Flinn et al., “Antibiotic-induced gut microbiome remodeling reduces neuroinflammation in traumatic brain injury,” Communications Biology (2026). DOI: 10.1038/s42003-026-09737-1.
This work was provided by Houston Methodist, underscoring their commitment to advancing interdisciplinary research at the intersection of neurology and gastroenterology.