Groundbreaking research jointly led by the Burnet Institute and WEHI has revealed the intricate mechanisms through which the human immune system combats Plasmodium vivax, the parasite responsible for the most prevalent type of malaria worldwide. Detailed in the prestigious journal Immunity, this comprehensive study delivers vital insights into the nature of protective immunity against P. vivax. It pinpoints precise targets on the parasite’s surface and elucidates the precise ways in which antibodies operate to both prevent initial infections and eliminate existing ones effectively.
Dr. Herber Opi, a Senior Research Fellow at the Burnet Institute, highlighted the current stagnation in worldwide efforts to control malaria, noting that despite significant advancements over many years, progress has recently plateaued. He emphasized that although two vaccines against malaria have been introduced in certain African regions, these interventions specifically address Plasmodium falciparum and provide absolutely no defense against P. vivax, which remains the predominant strain across vast areas of Asia and the Pacific region.
Overcoming Challenges in P. vivax Vaccine Development
One of the primary barriers hindering the creation of an effective vaccine for P. vivax has been the longstanding lack of clarity regarding the exact characteristics of protective immunity. Professor James Beeson, who serves as the Head of the Malaria Immunity and Vaccines program at the Burnet Institute, underscored the importance of these discoveries in shaping future vaccine strategies. He stated that these revelations unlock promising pathways for crafting vaccines tailored to P. vivax, which could substantially alleviate the global burden of malaria and facilitate efforts toward complete eradication of the disease.
Dr. Rhea Longley, Laboratory Head at WEHI, pointed out that the majority of international research initiatives and funding for malaria vaccines have been disproportionately directed toward P. falciparum, resulting in substantial gaps in our knowledge about P. vivax. She explained that P. vivax possesses distinct biological traits not found in P. falciparum, such as a dormant stage in the liver that leads to recurring relapses over time. This unique lifecycle complication renders it far more challenging to eradicate compared to other malaria species. Consequently, control measures proven effective against one parasite often fail to apply to the other, necessitating species-specific approaches.
Analyzing Immune Responses in High-Risk Populations
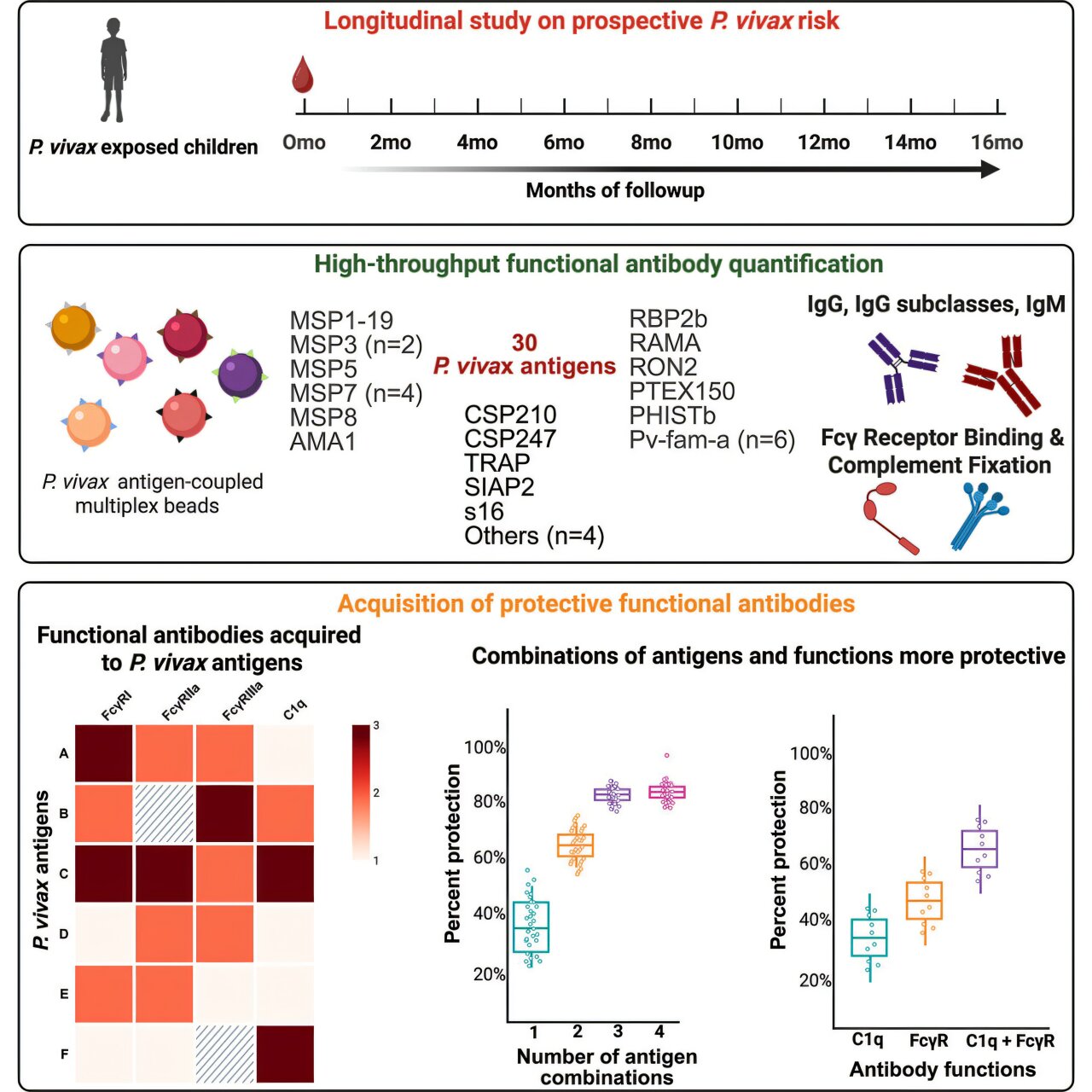
To delve deeper into these mechanisms, the research team utilized blood samples collected from children residing in Papua New Guinea, an area where P. vivax infections are particularly rampant. Through meticulous analysis, they investigated the interactions between antibodies and various components of the immune system, focusing on how these elements work together to avert the onset of clinical malaria symptoms.
The study’s results clearly demonstrate that protection against P. vivax does not simply correlate with the quantity or mere existence of antibodies in the bloodstream. Instead, the effectiveness hinges critically on the functional capabilities of these antibodies and the specific parasite proteins they are programmed to recognize and bind to. This nuanced understanding shifts the paradigm from a numbers-based view to a quality- and specificity-focused perspective on immunity.
Key Mechanisms of Effective Antibody Action
Investigators identified particular antibody responses that play a pivotal role in summoning immune cells to the site of infection and triggering essential immune signaling pathways designed to directly assault and dismantle the parasite. Notably, the potency of the immune response was markedly enhanced when antibodies targeted a diverse array of proteins simultaneously, rather than focusing on isolated targets. This multi-pronged attack amplifies the overall efficacy of the body’s defenses.
Furthermore, the research established that selecting the optimal combination of proteins for antibody targeting is of paramount importance. When antibodies effectively engage the correct set of multiple proteins, they can slash the risk of contracting malaria by more than 75 percent. This quantifiable outcome not only validates the approach but also furnishes a concrete blueprint for engineers and scientists working on next-generation vaccines.
Implications for Future Vaccine Innovation
These findings collectively represent a significant leap forward in the quest to develop the first truly effective vaccine against P. vivax. By mapping out the immune system’s blueprint for success against this elusive parasite, the study equips researchers with actionable data to prioritize vaccine candidates that mimic these naturally protective responses. The emphasis on functional antibody activities and multi-protein targeting strategies promises to accelerate development timelines and improve clinical outcomes in endemic regions.
In regions like Asia and the Pacific, where P. vivax accounts for the majority of malaria cases, such a vaccine could transform public health landscapes. It would not only reduce immediate infection rates but also interrupt transmission cycles, bringing the world closer to the long-term goal of malaria elimination. The collaborative nature of this research, bridging institutions like Burnet Institute and WEHI, exemplifies the power of pooled expertise in tackling complex global health challenges.
Looking ahead, ongoing longitudinal studies and clinical trials will be essential to translate these laboratory insights into real-world interventions. The detailed publication in Immunity ensures that these discoveries are accessible to the global scientific community, fostering further innovation and validation.
Publication and Study Details
The full study, titled “A longitudinal study of children identifies antibody Fc-mediated functions and antigen targets of immunity to Plasmodium vivax malaria,” was authored by D. Herbert Opi and colleagues. It appeared in Immunity in 2026, with a DOI of 10.1016/j.immuni.2026.02.003. This peer-reviewed work underwent rigorous scrutiny, ensuring its reliability and scientific integrity.