A Promising Advancement in ALS Treatment
A scientist from the University of Missouri has achieved a significant milestone in the effort to assist individuals suffering from amyotrophic lateral sclerosis (ALS), the progressive neurodegenerative condition also referred to as Lou Gehrig’s disease. Through a groundbreaking study, Mizzou researcher Smita Saxena demonstrated that a naturally occurring molecule known as GM1 can successfully penetrate the brain when encapsulated within minuscule lipid vesicles. Initial experiments conducted in laboratory settings revealed that this innovative technique substantially alleviated symptoms associated with ALS.
This development holds tremendous potential to initiate human clinical trials in the near future at the Roy Blunt NextGen Precision Health facility, where investigators aim to determine if this novel therapeutic strategy can enhance motor capabilities in ALS patients. The study, titled “Engineered GM1 intersects between mitochondrial and synaptic pathways to ameliorate ALS pathology,” appeared in the prestigious journal Advanced Science.
Understanding the Complex Neural Cascade in ALS
Treating ALS presents formidable challenges due to the multifaceted disruptions occurring within the brain, which progressively worsen and manifest as muscle weakness, speech difficulties, and profound exhaustion. These interconnected failures in neural function lie at the heart of the disease’s debilitating effects.
Smita Saxena, a distinguished professor in the School of Medicine at the University of Missouri, elaborated on prior findings from her research group: “Neurons affected by ALS exhibit heightened vulnerability to endoplasmic reticulum stress, which severely hampers the mitochondria’s capacity to generate essential energy. Consequently, this impairment restricts the neurons’ proficiency in transmitting signals across synapses, a critical process for coordinating muscle movements.”
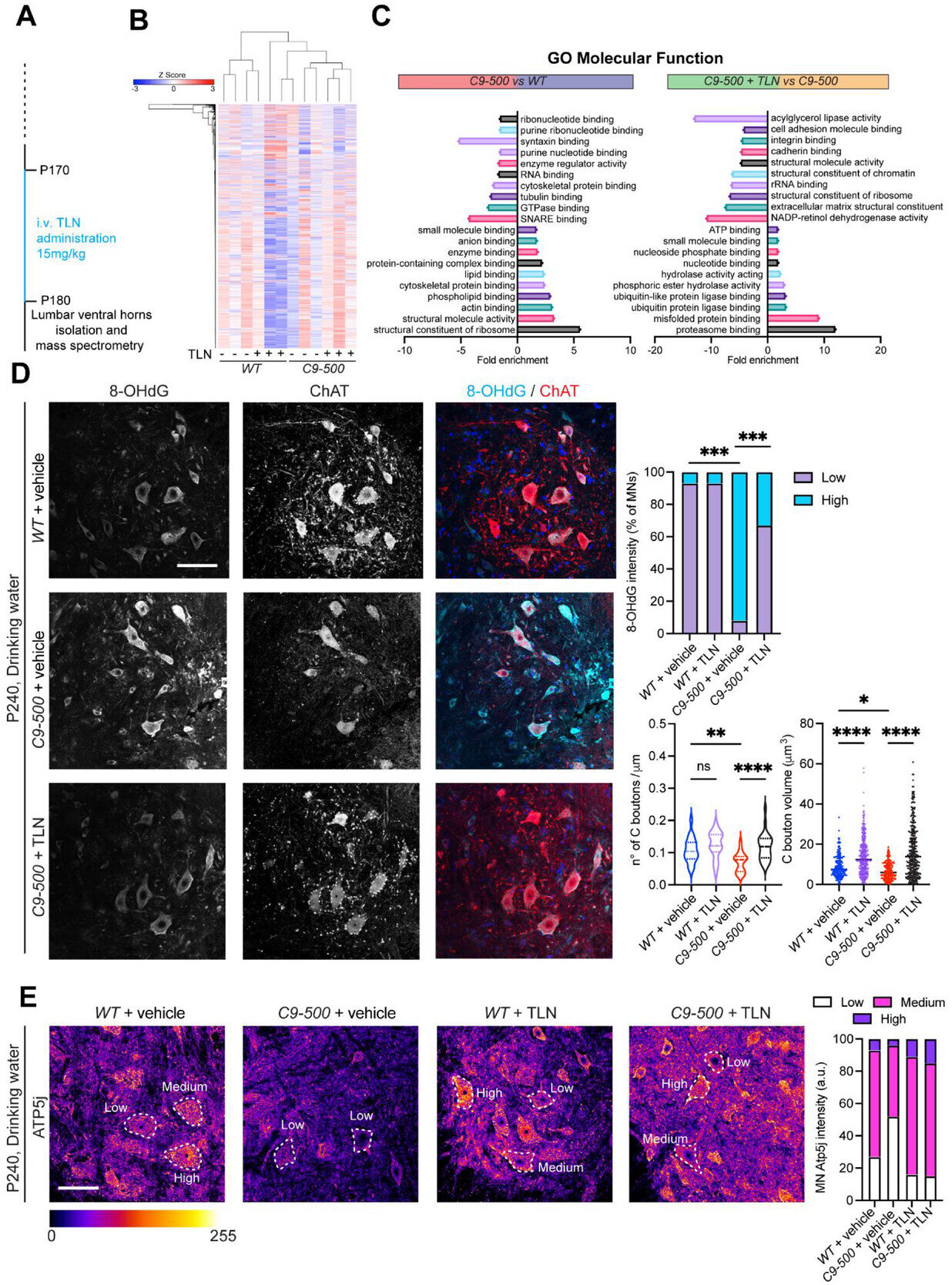
Such cascading neural malfunctions are the primary drivers behind the hallmark symptoms of ALS. Historically, efforts to transport effective ALS medications directly to the brain have been thwarted by the blood-brain barrier—a robust defensive shield composed of tightly packed cells that separates circulating blood from delicate brain tissue. While this barrier adeptly blocks pathogens and toxins, it regrettably obstructs beneficial therapeutics from accessing the central nervous system. As a result, earlier initiatives to supplement GM1 levels in the brain have consistently fallen short.
Saxena’s latest investigation appears to have surmounted this longstanding obstacle with remarkable ingenuity.
Revolutionary Talineuren Delivery Technology
Collaborating with InnoMedica, a pioneering biopharmaceutical firm based in Switzerland, Saxena employed a sophisticated delivery mechanism called Talineuren. This system consists of microscopic lipid nanoparticles that encase GM1, enabling safe and efficient transit across the blood-brain barrier. Notably, Talineuren has already undergone successful human clinical testing for delivering supplemental GM1 to individuals battling Parkinson’s disease, providing a proven safety profile that inspired Saxena to adapt it for ALS applications.
The results were exceptionally encouraging. Administered to mouse models engineered with the specific genetic mutation responsible for familial ALS, the Talineuren-GM1 formulation effortlessly breached the blood-brain barrier. It subsequently bolstered the vitality and functionality of motor neurons, leading to noticeable enhancements in the animals’ mobility and overall motor performance.
Should forthcoming clinical trials in humans validate these findings by demonstrating the therapy’s ability to decelerate ALS progression, medical professionals anticipate its potential administration to younger patients carrying the hereditary mutation. This proactive intervention could preemptively stall disease onset, preventing or delaying symptom appearance altogether. Adding to the optimism, the therapy’s established safety and excellent tolerability in Parkinson’s patients further bolsters confidence in its translational viability for ALS.
The Pivotal Role of Mizzou’s NextGen Precision Health Facility
Saxena credits the University of Missouri’s state-of-the-art infrastructure for enabling this critical research. “The Roy Blunt NextGen Precision Health building represents an ideal hub for such innovative studies,” she emphasized. “Integrating cutting-edge research laboratories with clinical trial spaces under one roof dramatically accelerates the journey from fundamental discoveries to practical human applications. This synergy promises to elevate the quality of life not only for Missouri residents but for ALS sufferers across the globe.”
Scientific Details and Broader Implications
The research underscores how engineered GM1 effectively bridges deficits in mitochondrial energy production and synaptic communication, two intertwined pathways ravaged by ALS pathology. By sustaining cholinergic synaptic inputs and preserving mitochondrial functionality in living models—as vividly illustrated in the study’s visualizations—the therapy addresses core disease mechanisms at their root.
Detailed publication information includes: Federica Pilotto et al., “Engineered GM1 Intersects Between Mitochondrial and Synaptic Pathways to Ameliorate ALS Pathology,” Advanced Science (2026). DOI: 10.1002/advs.202514128.
This work not only advances our comprehension of ALS neuropathology but also exemplifies how repurposing validated delivery platforms can unlock new therapeutic frontiers. The successful navigation of the blood-brain barrier via lipid nanoparticles like Talineuren may inspire similar strategies for other intractable neurological disorders, where drug delivery remains a persistent bottleneck.
Furthermore, the study’s emphasis on early intervention highlights a paradigm shift in neurodegenerative disease management—from reactive symptom palliation to preventive modulation. For patients with known genetic predispositions, this could mean transforming ALS from an inexorable decline into a manageable condition, markedly extending healthy lifespans and preserving independence.
As research progresses toward human trials, ongoing monitoring of long-term efficacy, dosage optimization, and potential off-target effects will be paramount. Yet, the foundational success in preclinical models, coupled with prior human safety data, positions this GM1-Talineuren approach as one of the most auspicious candidates in the ALS therapeutic pipeline.