Prolonged blood stagnation within the liver, referred to as chronic liver congestion, frequently results in a spectrum of severe conditions, including some that prove lethal. Nevertheless, the precise mechanisms by which liver congestion precipitates these illnesses have long eluded clear understanding, thereby complicating the identification of optimal therapeutic strategies. Researchers from Japan, in a comprehensive investigation detailed in the journal Gastroenterology, have successfully delineated a critical molecular cascade that links liver congestion directly to the onset of liver fibrosis, portal hypertension, and the development of liver tumors. These revelations carry substantial promise for the advancement of innovative treatment options.
Understanding Chronic Liver Congestion and Its Progression
Chronic liver congestion, alternatively termed congestive hepatopathy, is well-recognized for its tendency to evolve into liver fibrosis, which can advance to cirrhosis and ultimately hepatocellular carcinoma. Although these connections have been documented extensively in medical literature over many years, the underlying molecular processes that bridge liver congestion to fibrosis have remained largely enigmatic.
Determined to illuminate these gaps, a team of scientists from Osaka University embarked on an in-depth exploration of the relevant biological pathways, aiming to pinpoint viable therapeutic targets capable of halting the progression from liver congestion to more life-threatening hepatic disorders.
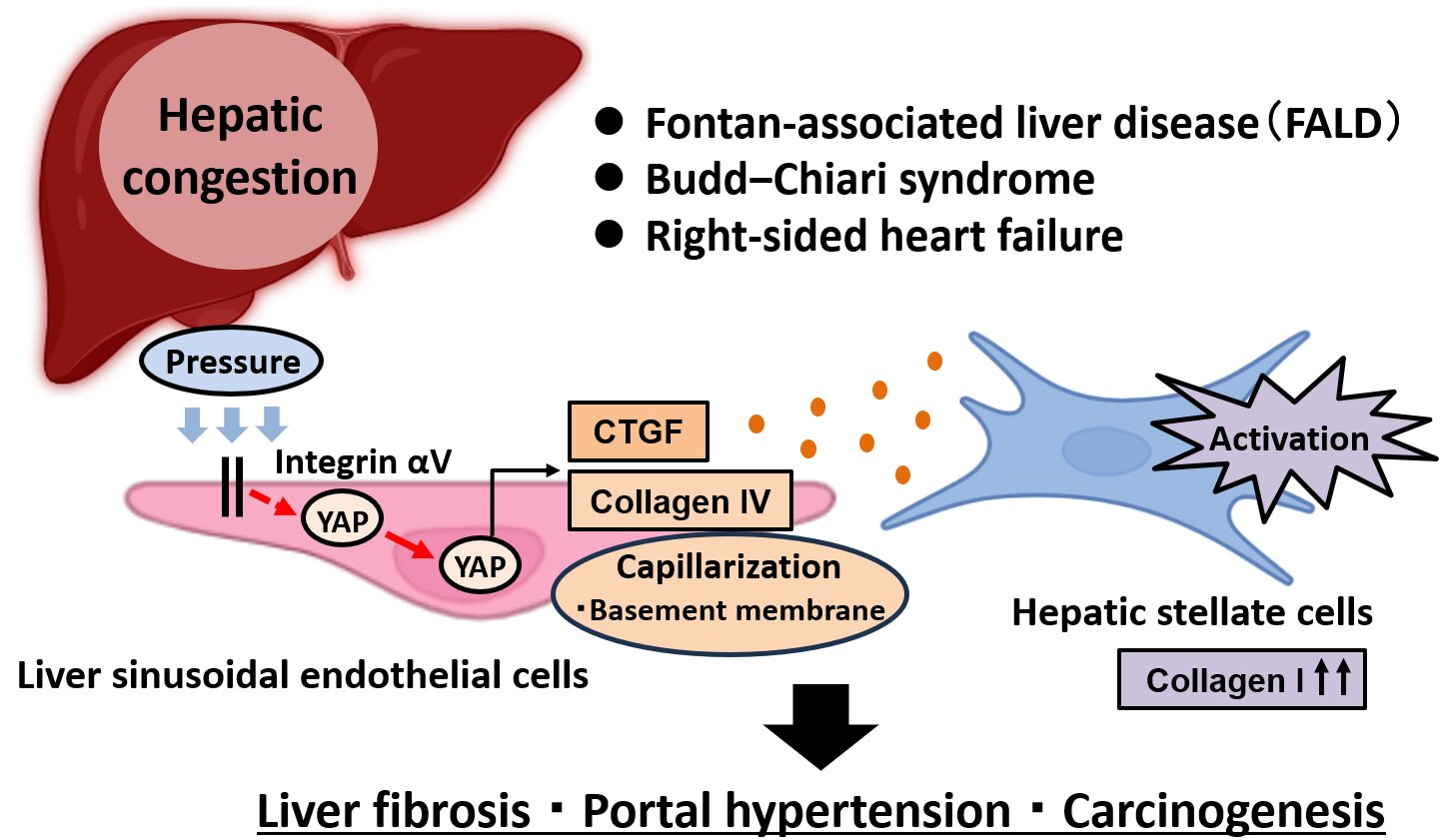
“Our investigation centered on liver sinusoidal endothelial cells (LSECs), the specialized cells that line the minuscule blood vessels within the liver and are particularly vulnerable when blood flow becomes obstructed or diminished, as is common in cases of liver congestion,” explained Seiya Kato, the study’s lead investigator.
“To probe these dynamics, we employed cutting-edge methodologies, including single-cell and spatial transcriptomics. These advanced tools enabled us to examine gene expression patterns at the level of individual cells and map their precise positions within liver tissues. We applied these techniques to samples obtained from a mouse model simulating congestion as well as human biopsies from individuals suffering from conditions like Fontan-associated liver disease. This approach allowed us to reveal the intricate molecular alterations triggered by liver congestion.”

Key Molecular Discoveries in LSECs
Through their meticulous analyses, the researchers identified heightened activity of two pivotal signaling molecules within LSECs: Yes-associated protein (YAP) and connective tissue growth factor (CTGF). Notably, activation of the integrin pathway was also evident in the congestion mouse model. By culturing LSECs in controlled laboratory settings, the team experimentally confirmed that elevated hydrostatic pressure—mimicking the conditions of chronic liver congestion—stimulates YAP activation via integrin αV, which in turn boosts CTGF expression.
Further experiments demonstrated that either pharmacologically blocking integrin αV or genetically eliminating CTGF in LSECs significantly ameliorated disease severity in the mouse congestion model.
Translating Findings from Mice to Human Patients
To assess the relevance of these observations in clinical contexts, the researchers conducted single-cell and spatial transcriptomic profiling on liver biopsies from human patients experiencing chronic liver congestion. Remarkably, they observed an identical molecular signature: YAP activation in LSECs correlated with elevated CTGF levels, patterns believed to drive pathological progression.
“In summary, we have uncovered a crucial signaling cascade—the integrin αV–YAP–CTGF pathway—operating within these unique liver vascular cells that mechanistically ties liver congestion to the development of fibrosis,” noted Hayato Hikita, the senior author on the study. “This novel pathway holds considerable potential as a foundation for future therapeutic interventions.”

Broader Clinical Implications and Potential Applications
Given the capacity of chronic liver congestion to precipitate grave complications such as fibrosis, portal hypertension, and primary liver cancer, these insights could profoundly influence patient care across diverse populations. A particularly relevant group includes individuals with congenital heart defects who have undergone the Fontan procedure, rendering them susceptible to congestion-induced hepatic injury.
Additionally, the heightened intravascular pressure characteristic of chronic liver congestion mirrors scenarios observed in advanced liver cirrhosis from various etiologies. Consequently, the mechanisms elucidated in this research may extend beyond congestion-specific hepatopathy, offering pathways for novel therapies applicable to cirrhosis patients irrespective of the precipitating cause.
By mapping this integrin αV–YAP–CTGF axis in liver sinusoidal endothelial cells, the study not only enhances our comprehension of congestive hepatopathy’s progression to fibrogenesis, portal hypertension, and carcinogenesis but also paves the way for targeted pharmacological strategies to interrupt these harmful processes early in their course.