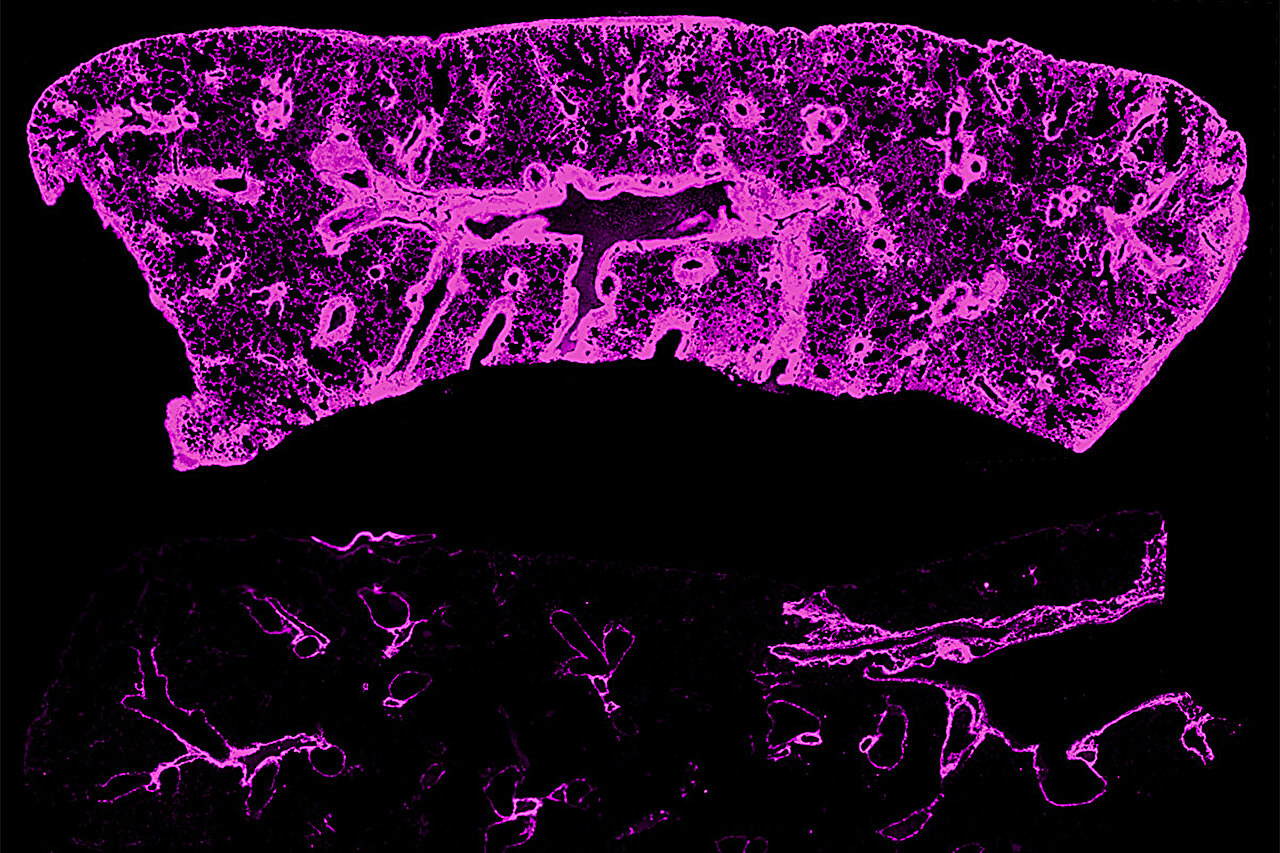
Researchers at Washington University School of Medicine have uncovered a key factor in chronic rejection of transplanted lungs and hearts: lymphatic vessels and their transported sugar molecule, hyaluronan, depicted in pink. In an image of a lung transplant from genetically identical mice, the upper section reveals extensive scar tissue loaded with hyaluronan. After applying a treatment to encourage lymphatic vessel regeneration, the accumulation of hyaluronan is substantially diminished in the lower section. Credit: Hailey Shepherd.
Although significant progress has been made in organ transplantation techniques, chronic rejection continues to pose a major challenge for patients, often emerging years or even a decade following heart or lung transplants. Traditionally, this persistent organ failure has been solely blamed on the recipient’s immune system progressively targeting the donor organ as foreign.
However, a groundbreaking investigation spearheaded by scientists at Washington University School of Medicine in St. Louis reveals that chronic rejection might actually originate from the interruption of lymphatic vessels—vital components of the body’s drainage network—within the donor organ, rather than direct immune assaults from the host.
Detailed in the journal Science Translational Medicine, the research encompasses examinations of human transplanted organs exhibiting chronic rejection alongside experimental mouse models for lung and heart transplants.
The findings indicate that impaired lymphatic drainage, which begins immediately upon the organ’s extraction from the donor, initiates a cascade of events culminating in fibrosis—a damaging condition where scar tissue gradually overtakes functional tissue in the graft. Moreover, the study demonstrates that halting the accumulation of sugar molecules typically cleared via lymphatic channels, or swiftly reinstating proper drainage post-transplant—potentially using medications already authorized for other ailments—can prevent fibrosis from developing.
Post-transplant, patients require lifelong immunosuppressive therapy to avert acute rejection. Nevertheless, chronic rejection frequently occurs despite this regimen, sometimes after extended periods of apparent stability. Clinicians may escalate immunosuppression in such instances, yet this approach typically fails, a longstanding enigma in transplantation medicine, notes senior author Daniel Kreisel, MD, Ph.D., holder of the G. Alexander Patterson, MD/Mid-America Transplant Endowed Distinguished Chair in Lung Transplantation at Washington University School of Medicine.
“Currently, our only recourse for chronic organ rejection is retransplantation, for which numerous patients do not qualify,” explains Kreisel, who also serves as a professor and vice chair for research in the Department of Surgery at Washington University School of Medicine. “This research is particularly thrilling as it identifies a novel, immune-independent mechanism behind chronic rejection, and our results suggest viable treatment options.”
In contrast to blood vessels, which surgeons meticulously reconnect during transplantation procedures, lymphatic vessels from the donor organ are left unlinked. These minuscule structures remain invisible even under surgical magnification, and it was previously assumed they naturally regenerated within a few weeks post-procedure.
“While regrowth does occur, our findings highlight that the initial two-to-three-week disruption phase post-transplant represents a crucial vulnerability period with lasting negative impacts on organ health,” states lead author Hailey Shepherd, MD, a surgical resident at Washington University School of Medicine who performed the experiments in Kreisel’s laboratory. “Fortunately, this same timeframe offers a strategic window for interventions to support lymphatic recovery and potentially avert the chronic deterioration observed in failing grafts.”
Tracking down the culprit
To pinpoint the roots of chronic rejection, Shepherd leveraged Washington University School of Medicine’s extensive biorepository of human organ specimens from patients with chronic rejection who underwent subsequent retransplants.
Analysis of these fibrotic tissues revealed a striking correlation: fibrotic regions precisely aligned with the lymphatic network. Further investigation showed that fibrosis in human samples stems from the pathological accumulation of hyaluronan, a sugar molecule. This buildup occurs in the graft due to compromised lymphatic drainage from severed vessels, ultimately driving the fibrosis synonymous with chronic allograft failure.
Utilizing mouse models of lung transplantation, the team explored three distinct interventions to eliminate hyaluronan from lymphatic vessels. The first inhibited the enzyme that synthesizes hyaluronan, maintaining vessel patency during the healing phase. The second promoted the proliferation of new lymphatic vessels, enhancing drainage capacity through increased vascular density. The third disrupted signaling pathways that prompt certain cells to overproduce hyaluronan.
Each method successfully prevented hyaluronan buildup and blocked chronic fibrosis in the mouse lung allografts. In several instances, the intervention-treated organs appeared virtually identical to non-transplanted healthy controls, as reported by the investigators.
Kreisel and Shepherd underscore that these transplants were performed between genetically identical mice, eliminating any possibility of alloimmune responses. This design confirms that fibrosis arises solely from the physical severance of lymphatic vessels, independent of adaptive immunity against donor antigens.
Although centered on lungs and hearts, the ubiquitous presence of lymphatic vessels across all solid organs implies that lymphatic obstruction might contribute to chronic rejection universally in transplantation.
One promising therapy, 4-methylumbelliferone (4-MU), inhibits hyaluronan synthesis and is already approved in Europe and Asia for biliary conditions affecting the gallbladder. The researchers highlight its strong safety record, positioning it as a prime candidate for clinical trials in transplant recipients.
Approaches to stimulate lymphatic vessel growth could be delivered directly to the organ pre- and peri-transplant, though their side effect profiles warrant further scrutiny. Meanwhile, targeting hyaluronan production signals requires cell-specific precision, presenting additional translational hurdles.
Beyond post-transplant administration to recipients, these therapies hold potential for ex vivo treatment of organs during preservation in cold storage solutions prior to implantation. Unlike perpetual immunosuppression, lymphatic-focused interventions could be finite, ceasing once drainage functionality is reestablished.
“Our immediate priority is determining the optimal pathway to clinical implementation for the benefit of transplant patients,” Kreisel affirmed.