Human metabolism operates under the influence of internal biological clocks known as circadian rhythms. Numerous physiological processes, including the absorption of sugars, the oxidation of fats, and the secretion of hormones, display daily fluctuations that follow distinct 24-hour patterns. When individuals ingest nutrients outside these ideal temporal windows aligned with their circadian cycles, it may contribute to the emergence of metabolic disturbances over extended periods. Chrononutrition, as a scientific discipline, delves into the molecular underpinnings of these effects to refine eating schedules, thereby enhancing overall metabolic wellness and helping to avert chronic illnesses.
Fats in the Morning or Evening: What Makes the Difference?
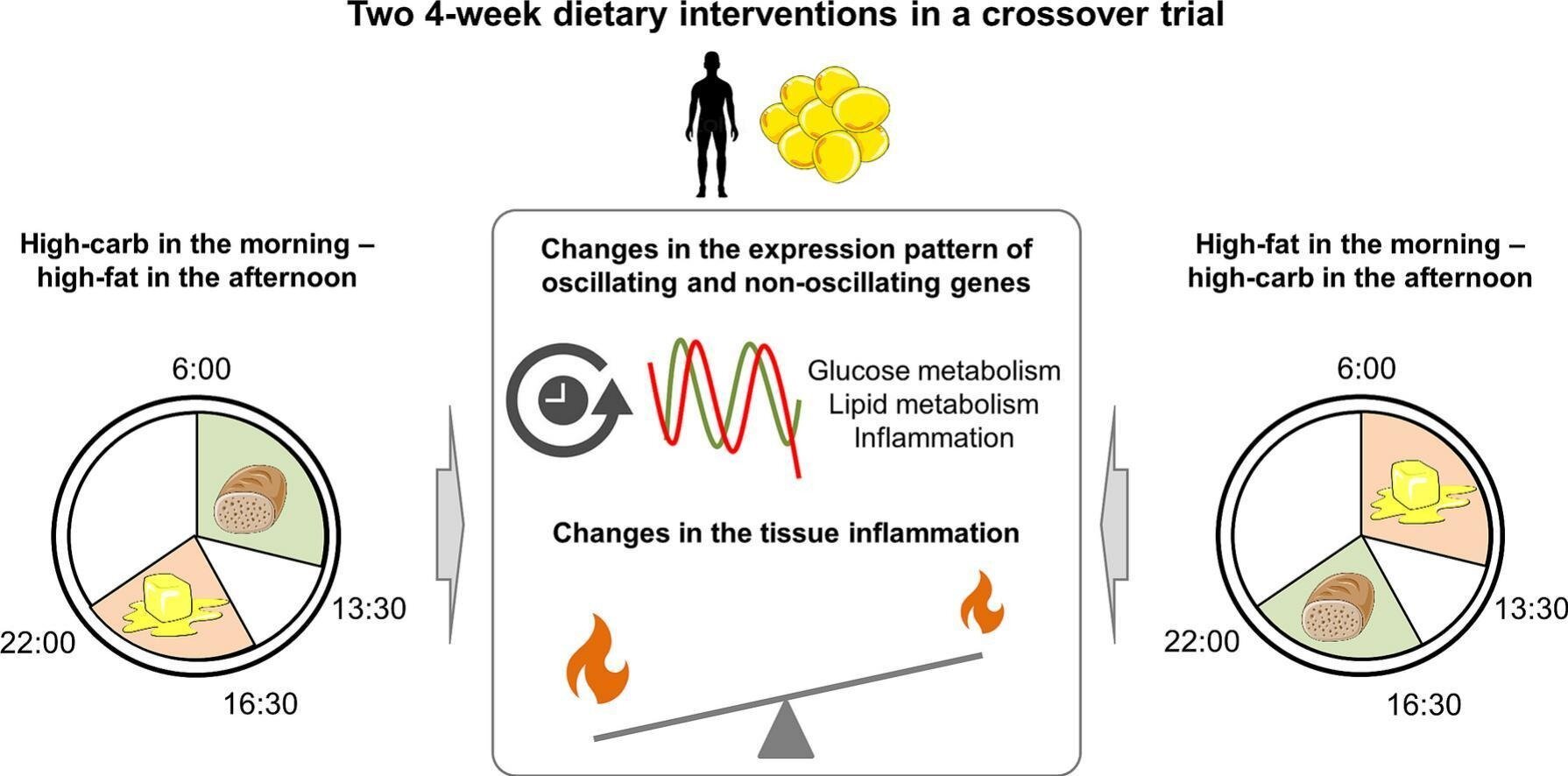
Professor Olga Ramich and her research group at the German Institute of Human Nutrition Potsdam-Rehbruecke (DIfE) have spearheaded an innovative human trial to determine if the daily timing of carbohydrate and fat consumption impacts gene expression within subcutaneous adipose tissue, independent of total caloric consumption. This investigation drew upon information from a meticulously controlled dietary experiment employing a crossover methodology, featuring 29 overweight male participants who did not have diabetes. Each participant adhered to two distinct nutritional regimens, both equated in terms of energy content, over separate four-week periods.
In one regimen, carbohydrates were emphasized during the morning meal, with fats concentrated in the evening. The alternative regimen inverted this sequence. Such a structured approach enabled the scientists to more precisely discern personalized metabolic reactions by minimizing confounding variables.
Throughout the study, the researchers obtained biopsy samples from the subcutaneous fat deposits of the participants at three specific intervals on the assessment day—both prior to and following each dietary phase. Working in collaboration with Professor Achim Kramer, who leads the Chronobiology Research Area in the Department of Anesthesiology and Intensive Care Medicine at Charité, the team examined the transcriptome of these samples. The transcriptome provides a comprehensive, real-time profile of which genes are actively being transcribed and to what extent, offering deep insights into cellular activity.
Distinct Patterns Emerge in Fat Tissue Responses
Marking a significant milestone, Ramich’s team has, for the first time, meticulously mapped the rhythmic gene expression profiles in human subcutaneous adipose tissue. They conclusively showed that varying the timing of macronutrient intake throughout the day modifies both the rhythmic and arrhythmic components of the transcriptome. These groundbreaking results have been detailed in the esteemed journal Food Research International.
Detailed genomic analyses revealed that as many as 1,386 genes in the studied fat tissue display diurnal oscillations, meaning their activity levels rise and fall predictably over the course of a day. A substantial portion of these rhythmically expressed genes plays crucial roles in glucose metabolism, lipid processing, or the modulation of inflammation. Notably, the timing of macronutrient ingestion exerted a profound effect on the daily expression patterns of approximately one-third of these oscillating genes within the adipose tissue.
Certain genes experienced shifts in their oscillation characteristics, such as changes in peak timing or amplitude. In other instances, genes that previously oscillated lost their rhythmicity, while some non-oscillating genes newly acquired daily rhythms in response to the dietary timing manipulations.
Particularly striking were the outcomes related to insulin sensitivity indicators in the adipose tissue. When participants consumed a diet higher in fats during the morning and richer in carbohydrates during the evening, there was a measurable enhancement in these insulin sensitivity markers. In contrast, relocating fat consumption to the evening hours resulted in heightened expression of genes linked to inflammation. This pattern suggests the onset of a pro-inflammatory environment within the fat tissue, which is widely recognized as a precursor to metabolic pathologies including excessive weight gain and type 2 diabetes mellitus.
Pioneering Strategies for Metabolic Disease Prevention
Professor Ramich elaborates, “Our findings substantiate preliminary evidence indicating that altering the distribution of macronutrients across the day produces detectable changes in the metabolic functions of adipose tissue. Consuming meals laden with fats later in the day might initiate adverse molecular cascades that foster inflammation and metabolic dysregulation with prolonged exposure.”
The research team views these discoveries as opening promising pathways for the prophylaxis of obesity and type 2 diabetes. Ramich further notes, “Beyond merely controlling caloric intake and selecting appropriate nutrient profiles, the chronological placement of meals could emerge as a vital element in tailoring individualized nutrition plans moving forward.”
That said, the investigators stress the necessity for more expansive, prolonged intervention trials to substantiate health outcomes and formulate precise guidelines applicable to diverse demographics, including females and individuals diagnosed with diabetes. Complementary mechanistic investigations utilizing in vitro cell cultures or in vivo animal models would further illuminate the interplay between circadian alterations in adipose tissue dynamics and broader systemic metabolic regulation.
This study underscores the intricate interplay between our daily eating habits and the molecular clockwork governing fat tissue function. By highlighting how nutrient timing can reprogram gene activity, it lays the groundwork for chrononutrition-based interventions that could significantly bolster public health efforts against the rising tide of metabolic disorders. Future research will undoubtedly build upon this foundation, exploring dose-response relationships, genetic predispositions, and lifestyle synergies to craft even more effective preventive strategies.
In essence, the research illuminates the profound sensitivity of adipose tissue’s genetic machinery to the diurnal patterning of dietary macronutrients. This sensitivity manifests not just in shifts to existing rhythms but in the induction or suppression of oscillatory behavior altogether, with direct implications for metabolic homeostasis and inflammatory control. The observed improvements in insulin responsiveness from morning fat loading suggest an alignment with endogenous circadian peaks in lipid metabolism, potentially optimizing energy partitioning and storage efficiency.
Conversely, evening fat enrichment appears to desynchronize these processes, tipping the balance toward inflammatory signaling pathways that could erode tissue health over time. These insights challenge conventional nutritional dogma focused solely on ‘what’ we eat, compelling a reevaluation to incorporate ‘when’ as a critical variable in dietary therapeutics. As personalized medicine advances, integrating chronobiological principles into nutrition counseling holds immense promise for mitigating cardiometabolic risks in at-risk populations.