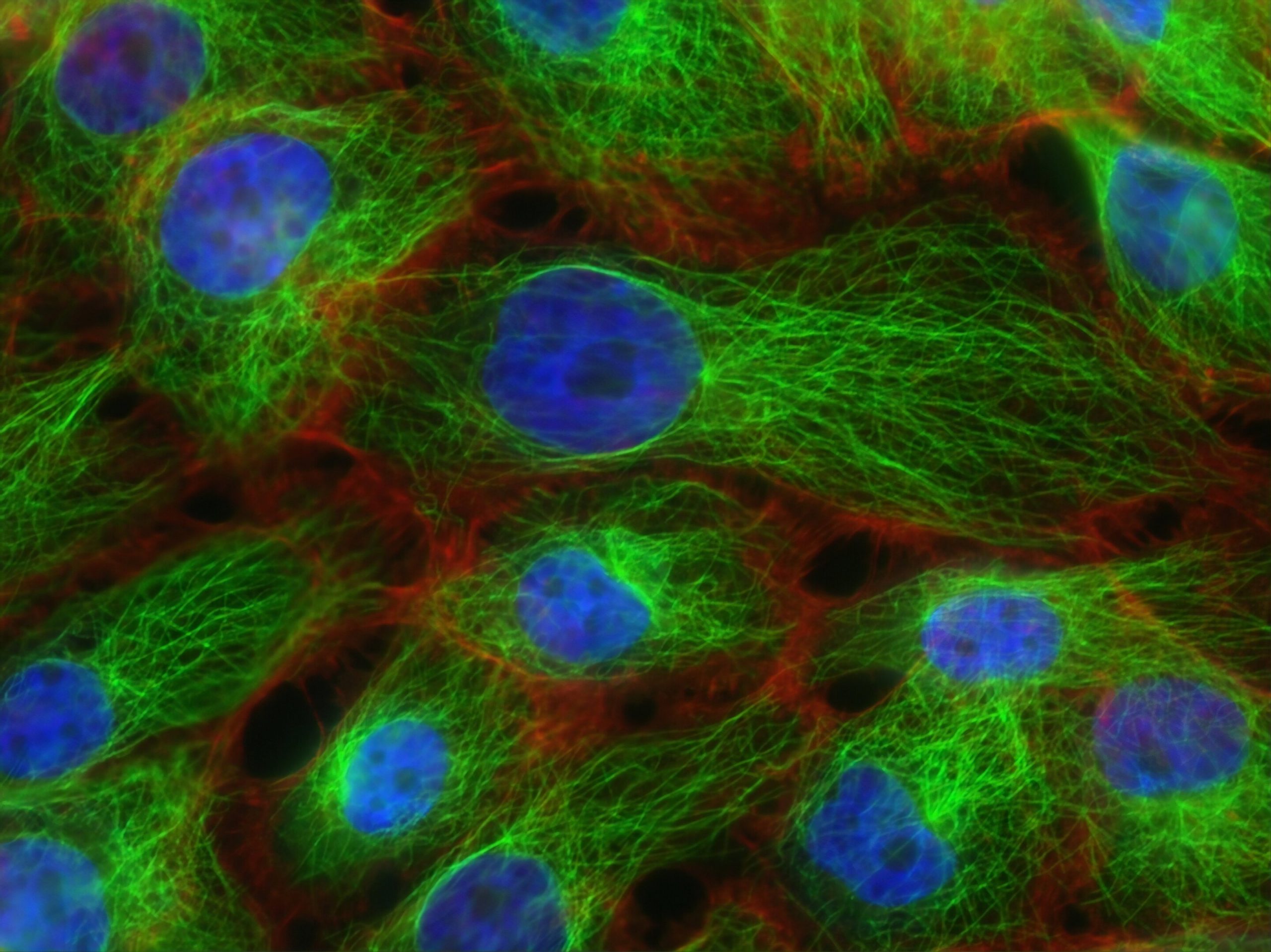
Approximately 50% of patients diagnosed with triple-negative breast cancer (TNBC) eventually develop resistance to their prescribed therapies. This resistance significantly heightens the risk of tumor recurrence following initial treatment, thereby drastically diminishing overall survival prospects for those affected. A team of scientists from Baylor College of Medicine has uncovered compelling evidence indicating that TNBC employs more than a single strategy to evade therapeutic interventions.
The groundbreaking research, detailed in the esteemed Journal of Clinical Investigation, delineates two distinct and mutually exclusive mechanisms through which TNBC acquires the capacity to withstand treatment. These revelations carry profound implications for clinical practice, as anticipating the specific resistance pathway a patient’s tumor might activate could enable physicians to tailor interventions proactively, thereby mitigating or altogether averting resistance development.
Diverse Resistance Mechanisms in Triple-Negative Breast Cancer
Insights gleaned from this investigation, building upon prior work conducted within the same laboratory, underscore the multifaceted nature of therapy resistance in TNBC. “Our findings, combined with those from an earlier project in our group, demonstrate unequivocally that TNBC can forge resistance through multiple avenues,” explained Dr. Xiang Zhang, the study’s lead author. Dr. Zhang holds the position of professor and occupies the William T. Butler, M.D., Endowed Chair for Distinguished Faculty in the Department of Molecular and Cellular Biology. He also directs the Lester and Sue Smith Breast Center and serves as a key member of the Dan L. Duncan Comprehensive Cancer Center, all affiliated with Baylor College of Medicine.
It is crucial to recognize that no two TNBC tumors are identical in their makeup. Each exhibits a unique blend of tumor cells interspersed with various immune cells, including macrophages and neutrophils. The precise cellular architecture of a given tumor dictates the trajectory it will pursue toward therapeutic resistance.
To elucidate these resistant pathways, the research team meticulously analyzed tissue samples obtained directly from patients alongside sophisticated mouse models that recapitulate human TNBC. In a preceding investigation, they had established that within epithelial-like TNBC variants—characterized by the presence of both macrophages and neutrophils—it is predominantly the neutrophils that orchestrate resistance against therapeutic agents.
Shifting Roles of Immune Cells in Tumor Resistance
However, the latest study unveils a nuanced shift: neutrophils do not universally spearhead resistance. In a contrasting subtype known as mesenchymal-like TNBC tumors, which are predominantly populated by macrophages, these macrophage cells emerge as the primary architects of resistance.
“Our experiments revealed that chemotherapy possesses the ability to reprogram these macrophages, effectively converting them from vigilant defenders of the body into accomplices that shield tumors from immune-mediated destruction,” noted Dr. Liqun Yu, a co-first author and postdoctoral researcher in Dr. Zhang’s laboratory. “These altered macrophages actively phagocytose—or engulf and digest—cancer cells, yet they simultaneously secrete an array of immunosuppressive compounds, such as C1q and resolvin. These substances actively dampen the body’s natural immune assault on the malignancy.”
Delving deeper into potential countermeasures, the scientists evaluated multiple interventions aimed at neutralizing the detrimental influence of these reprogrammed, suppressive macrophages within mesenchymal-like tumors. Strategies including the complete depletion of macrophages or pharmacological blockade of their infiltration into the tumor microenvironment successfully reinstated the tumors’ vulnerability to standard treatments. Furthermore, inhibiting the production of key immunosuppressive factors secreted by these macrophages revitalized the antitumor immune response, paving the way for innovative tactics to forestall the onset of therapy resistance.
Implications for Personalized Cancer Treatment
These discoveries illuminate a promising frontier in oncology: the prospect of preemptively forecasting a tumor’s resistance trajectory based on its pretreatment cellular profile. “By scrutinizing the immune and tumor cell composition of a patient’s TNBC prior to initiating therapy, we could implement targeted preventive measures against resistance. This proactive stance holds the potential to markedly enhance survival rates,” Dr. Zhang emphasized.
The study meticulously delineates how inflammation- and resolution-programmed myeloid circuits—encompassing neutrophils and macrophages—differentially govern therapeutic outcomes in epithelial versus mesenchymal subtypes of TNBC. This duality underscores the imperative for subtype-specific therapeutic enhancements, potentially revolutionizing how clinicians approach this aggressive malignancy.
In epithelial-like tumors, neutrophils dominate the resistance landscape by fostering an immunosuppressive niche that shields cancer cells from chemotherapeutic annihilation. Conversely, mesenchymal-like tumors leverage macrophage reprogramming, where chemotherapy inadvertently flips these cells into tumor-promoting entities. This reprogramming cascade involves not only direct engulfment of dying cancer cells but also the orchestrated release of mediators like C1q, a component of the complement system, and resolvin, a lipid mediator renowned for resolving inflammation but here subverted to quell adaptive immunity.
The therapeutic countermeasures tested were both elegant and effective. Macrophage depletion via clodronate liposomes or genetic ablation in mouse models restored therapy sensitivity, as did CCR2 inhibitors that curtailed macrophage recruitment. Similarly, antagonists targeting C1q or resolvin pathways reinvigorated T-cell mediated cytotoxicity, suggesting a combinatorial regimen where standard chemotherapy is augmented with myeloid-targeted agents.
From a clinical perspective, this work advocates for routine tumor microenvironment profiling—perhaps via single-cell RNA sequencing or multiplex immunohistochemistry—to stratify patients into epithelial-like or mesenchymal-like categories pre-treatment. Such precision could guide the adjunctive use of neutrophil or macrophage modulators, transforming empirical therapy into a predictive, personalized paradigm.
Moreover, these findings resonate with broader insights into the tumor-immune interplay. They affirm that resistance is not merely a cancer cell-intrinsic phenomenon but a symphony orchestrated by the stromal and myeloid compartments. Prior research from the lab had spotlighted fatty acid-mediated immune suppression, linking metabolic rewiring to neutrophil dominance in certain contexts—a thread that weaves seamlessly into this narrative of cellular heterogeneity.
Looking ahead, clinical trials validating these biomarkers and interventions are eagerly anticipated. If successful, they could shatter the therapeutic ceiling for TNBC, a subtype long notorious for its dismal prognosis and paucity of targeted options. By dismantling these myeloid-mediated barricades, oncologists may finally tip the scales toward durable remission and improved longevity for patients battling this formidable foe.
Publication specifics include: Liqun Yu et al., ‘Inflammation- and resolution-programmed myeloid circuits govern therapeutic resistance in epithelial and mesenchymal triple-negative breast cancer,’ Journal of Clinical Investigation (2026). DOI: 10.1172/jci198815.
This comprehensive exploration not only demystifies the elusive mechanisms of TNBC resistance but also equips the medical community with actionable intelligence to combat it head-on, fostering hope for more efficacious strategies in the relentless war against cancer.