Researchers from the Nanobiotechnology for Diagnostics group (Nb4D) at the Institute of Advanced Chemistry of Catalonia (IQAC), which operates under the Spanish National Research Council (CSIC), have engineered a specialized monoclonal antibody. This innovative protein has proven effective in laboratory cell cultures, where it successfully neutralizes the potent toxin known as pyocyanin. This toxin is secreted by the bacterium Pseudomonas aeruginosa, a pathogen flagged by the World Health Organization as extremely hazardous. The bacterium’s notorious resistance to virtually all existing antibiotics, combined with its frequent occurrence in healthcare facilities, makes it a significant threat to patient safety.
The findings from this groundbreaking research appear in the esteemed journal ACS Pharmacology and Translational Science. The scientific team adopted a forward-thinking “anti-virulence” methodology in their work. Rather than directly attacking the bacterial cells as traditional antibiotics do, this novel strategy zeroes in on disarming the pyocyanin toxin specifically. This targeted approach carries the substantial benefit of sidestepping the mechanisms that typically foster the evolution of antibiotic resistance in pathogens.
Among the most alarming infectious agents on a global scale, Pseudomonas aeruginosa stands out due to its remarkable adaptability. It swiftly evolves defenses against the majority of antibiotics currently in clinical use, leading to countless fatalities annually, particularly among immunocompromised individuals in medical environments. The pyocyanin toxin is central to its destructive prowess, as it systematically destroys key immune cells and severely impairs the host’s natural inflammatory defenses. By counteracting this toxin, scientists are exploring a viable alternative treatment pathway that could dramatically lessen reliance on broad-spectrum antibiotics and mitigate the ongoing crisis of emerging resistant strains.
Monoclonal Antibody Development
Lluïsa Vilaplana, a prominent researcher at IQAC-CSIC and the principal investigator behind this study, underscores the pressing need for such paradigm-shifting interventions. She notes, “The bacterium’s extraordinary capacity for adaptation has rendered conventional antibiotics largely ineffective, compelling us to pioneer fresh therapeutic avenues. These innovations aim not only to combat multidrug-resistant variants but also to halt the advancement of infections at their core.” The relentless march of resistance elevates the quest for non-traditional solutions to an imperative in modern infectious disease management.
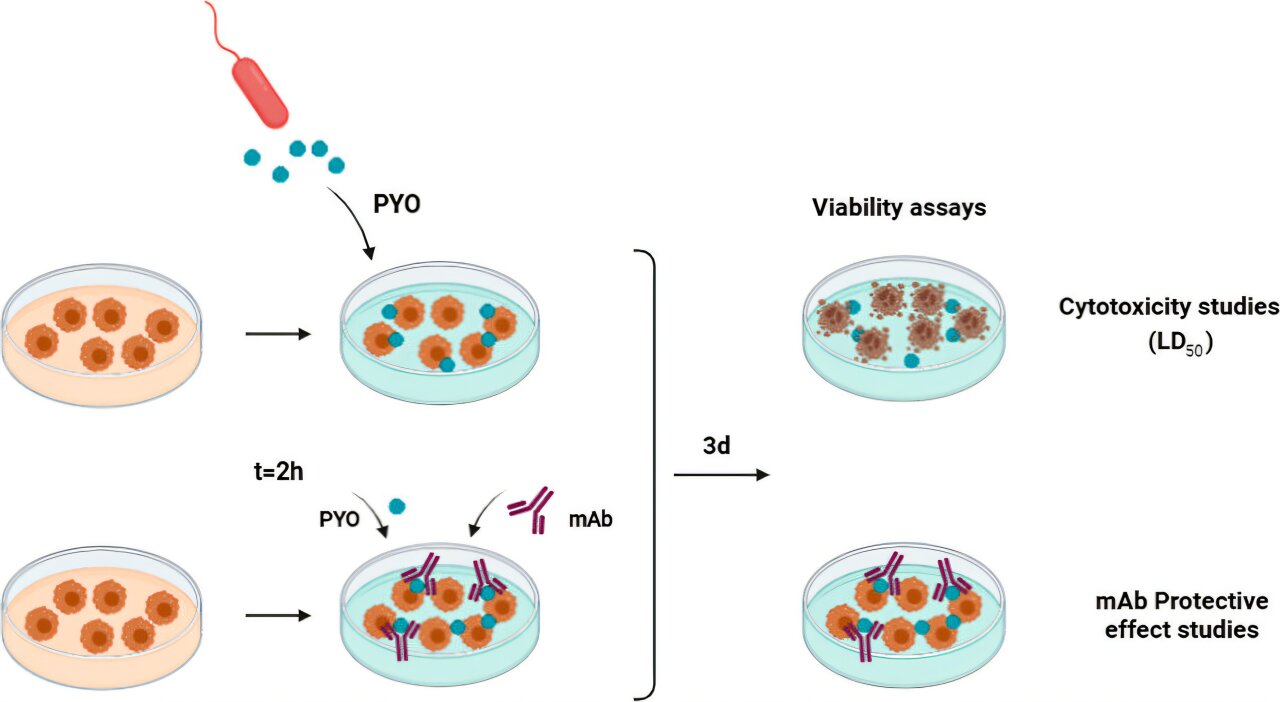
To tackle this challenge head-on, the IQAC team meticulously developed a monoclonal antibody dubbed mAb122 using mouse models. This precision-engineered protein is tailored to latch onto pyocyanin with exceptional specificity, thereby halting its toxic activities. Monoclonal antibodies, produced in controlled laboratory settings, excel at identifying and binding to unique molecular targets, enabling highly selective inhibition without collateral effects on surrounding biological processes.
Following successful production, the researchers rigorously tested mAb122 on macrophage cell lines—the vital immune cells often targeted by infections—after exposing them to varying doses of the pyocyanin toxin. The outcomes were compelling: the antibody markedly diminished the toxin’s capacity to inflict cellular harm and substantially boosted the survival rates of these critical immune defenders. Equally important, when applied independently, mAb122 exhibited no inherent toxicity, a crucial green light for advancing it toward clinical applications.
Pilar Marco, the director of the Nb4D group at IQAC and overseer of the project, emphasizes the transformative potential of this method. “This diverges sharply from standard antibiotic protocols, which seek to eradicate the pathogen outright,” she explains. “Instead, our anti-virulence tactic neutralizes a primary weapon in the bacterium’s arsenal, thereby alleviating the evolutionary pressures that drive resistance. It’s akin to stripping the invader of its most lethal tools rather than engaging in a direct confrontation.”
By focusing on virulence factors rather than bacterial survival, this antibody therapy promises to curtail the overuse of antibiotics, potentially allowing for reduced dosages in combination regimens. Such a shift avoids imposing selective pressures on bacterial populations, which are the root cause of resistance development. The strategy empowers the body’s innate defenses to manage the infection more effectively, preserving the efficacy of existing antimicrobial agents for truly dire circumstances.
In addition to its protective effects on cells, the investigation delved into the antibody’s influence on the inflammatory cascade. Pyocyanin is known to dysregulate cytokine production—signaling molecules that orchestrate immune responses—leading to excessive or misguided inflammation. The antibody did alter certain cytokine profiles, hinting at modulatory capabilities. However, these preliminary observations necessitate deeper exploration to fully characterize its immunological footprint and optimize its balance between protection and inflammation control in subsequent phases of research.
Ultimately, deploying antibodies to neutralize pyocyanin offers a compelling pathway for safeguarding immune cells at the frontline of defense against Pseudomonas aeruginosa‘s chief virulence agent. This method does not aim for bacterial eradication but rather curtails the pathogen’s destructive potential, enabling the immune system to mount a more robust counteroffensive. In clinical contexts, this could prove instrumental in averting life-threatening infections while curbing the antibiotic arms race.
While promising, the research remains in its nascent in vitro phase, setting the stage for essential in vivo validations in animal models. These upcoming trials will scrutinize the antibody’s safety profile, pharmacokinetic behavior, and therapeutic efficacy within living organisms. Should these experiments corroborate the cell culture data, mAb122 could emerge as a precision instrument in the fight against multidrug-resistant infections, particularly those rampant in hospitals. Future milestones include assessing its performance in preclinical animal studies and fine-tuning to preclude any adverse inflammatory repercussions.
Publication and Broader Implications
The detailed study, titled “Anti-pyocyanin Antibody Exhibits Cytotoxicity Protective Effects on Macrophages: A Promising Innovative Therapeutic Approach for Pseudomonas aeruginosa Infections,” was authored by Bárbara Rodríguez-Urretavizcaya and colleagues. It was published in ACS Pharmacology & Translational Science in 2025, with DOI: 10.1021/acsptsci.5c00187.
This work illuminates critical medical concepts such as the pathogenesis of Pseudomonas aeruginosa and the modulation of inflammation in bacterial infections. It falls within the domains of infectious diseases and clinical pharmacology, addressing a pivotal gap in treatment options for hospital-acquired pathogens.
Provided by the Spanish National Research Council, this advancement signals a hopeful trajectory for anti-virulence therapies. As antibiotic resistance continues to escalate, innovations like mAb122 could redefine infection management, prioritizing host protection and microbial disarmament over outright elimination. Ongoing research will be vital to translate these laboratory triumphs into bedside realities, potentially saving lives in high-risk healthcare settings worldwide.