A groundbreaking investigation conducted by scientists at the Hebrew University of Jerusalem sheds new light on the initial stages of pancreatic cancer development, revealing that this aggressive disease might start evading the body’s immune defenses much sooner than previously anticipated. The research demonstrates that precancerous cells in the pancreas begin to form specialized spatial compartments, known as niches, and initiate deliberate communications with nearby immune cells. These early interactions could foster an environment that suppresses immune responses right from the outset of the pathological process. The findings have been detailed in the prestigious journal Gastroenterology.
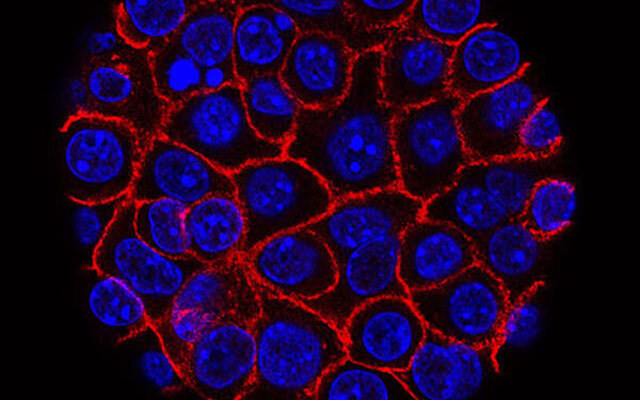
This comprehensive study was spearheaded by Dr. Oren Parnas, with significant contributions from graduate student Sebastian Arcila-Barrera and Dr. Sharona Tornovsky-Babeay, all affiliated with the Faculty of Medicine at the Hebrew University of Jerusalem. The team employed advanced methodologies, including single-cell RNA sequencing and spatial transcriptomics, to meticulously analyze samples of pancreatic tissue. These techniques allowed the preservation of the precise positional relationships among thousands of individual cells, enabling a detailed mapping of how various acinar metaplastic cells arrange themselves within premalignant formations and engage with adjacent immune components.
Pancreatic ductal adenocarcinoma stands out as one of the most lethal types of malignancy, primarily because it is frequently diagnosed at advanced stages when therapeutic interventions are severely limited. Precancerous abnormalities in the pancreas can persist for up to a decade or longer prior to the emergence of fully invasive tumors, yet the scientific community has historically possessed limited knowledge regarding the structural organization of these initial cellular alterations or their impact on the surrounding tissue microenvironment.
“Our research indicates that these initially transformed cells do not scatter haphazardly throughout the tissue,” explained Dr. Parnas. “Rather, they group into semi-uniform niches based on shared characteristics, and these clusters seem to purposefully engage with particular subsets of immune cells.”
The analysis uncovered that specific metaplastic cell configurations are repeatedly positioned in immediate vicinity to immune cells notorious for their suppressive roles, such as certain neutrophil variants and macrophage populations. These close associations correlated with distinct gene expression profiles that are recognized for inhibiting immune functions, thereby implying that mechanisms of immune avoidance could activate substantially before the cancer transitions to an invasive state.
Sebastian Arcila-Barrera highlighted the significance of these spatial arrangements in understanding the trajectory of the disease. “The patterns of cell distribution we documented point toward an early establishment of cellular identities, succeeded by targeted proliferation in localized areas,” he elaborated. “Such observations provide crucial insights into the mechanisms driving the formation and progression of premalignant lesions over extended periods.”
Dr. Sharona Tornovsky-Babeay underscored the potential clinical applications of these discoveries. “By gaining a deeper comprehension of how these lesions originate and mature, we stand to improve our ability to pinpoint those at highest risk for malignant transformation,” she stated. “Looking ahead, this knowledge could pave the way for innovative preventive measures that disrupt cancer development at its infancy, well before it manifests clinically.”
A key strength of the study lies in its validation across species: the researchers identified comparable patterns of cellular clustering and immune cell engagements in human pancreatic specimens, affirming the broader applicability of their observations beyond preclinical models. This cross-species consistency bolsters confidence in the translational potential of the results.
Collectively, these revelations paint a vivid portrait of the nascent phases of pancreatic cancer onset, emphasizing the critical roles played by spatial cellular architecture and early immune modulations in dictating long-term disease progression—often years prior to the appearance of any noticeable symptoms. Such early events could represent pivotal windows for therapeutic disruption.
Publication Information
The study, titled “Acinar Metaplastic Cells Generate Semi-homogeneous Niches and Interact with Immune Cells,” was published in Gastroenterology in 2026 (DOI: 10.1053/j.gastro.2025.12.014).
Broader Context and Implications
Pancreatic ductal adenocarcinoma’s notoriety stems not only from its rapid progression but also from its proficiency at concealing itself from immune surveillance. Traditional views posited that immune evasion tactics were predominantly a feature of established tumors, but this research challenges that paradigm by demonstrating immunosuppressive dynamics at the precancerous stage. The formation of semi-homogeneous niches—where like-minded metaplastic cells congregate—suggests a coordinated strategy that predates overt malignancy.
Single-cell RNA sequencing provided granular data on transcriptional states, while spatial transcriptomics added the vital dimension of location, revealing non-random distributions. For instance, acinar cells undergoing metaplasia, which adaptively change form in response to stress or injury, preferentially neighbor suppressive immune effectors. Neutrophils and macrophages in these niches express genes like those encoding arginase or PD-L1, classic dampeners of T-cell activity.
- Precancerous lesions persist for 10+ years before invasion.
- Metaplastic cells cluster into niches with immune suppressors.
- Gene signatures indicate early immune dampening.
- Patterns consistent in mouse models and human tissues.
- Potential for early detection and intervention strategies.
This spatial precision implies that disease evolution is not merely stochastic but architecturally orchestrated. Early niche formation might confer survival advantages, allowing mutated cells to expand unchecked. Immune cells, rather than mounting a defense, become unwitting accomplices in fostering tolerance.
For clinicians, these insights herald possibilities for refined risk stratification. Biomarkers reflecting niche-immune signatures could flag high-risk lesions via imaging or liquid biopsies. Therapeutically, targeting these nascent interactions—perhaps with checkpoint inhibitors or niche-disrupting agents—might halt progression preemptively.
The Hebrew University team’s rigorous approach, blending cutting-edge omics with histopathological validation, sets a new standard for studying early carcinogenesis. As pancreatic cancer claims lives silently, unraveling its covert beginnings offers hope for turning the tide through timely interventions.