Immunotherapy treatments have historically shown limited success against prostate cancer primarily because these tumors are classified as immunologically ‘cold.’ This designation indicates that they fail to draw sufficient immune cells to launch an effective assault on the malignancy. The standard hormone therapy employed for prostate cancer, known as androgen deprivation therapy (ADT), can briefly enhance tumor responsiveness by recruiting immune cells into the cancerous area. However, this advantage proves fleeting, as the therapy simultaneously elevates the presence of regulatory T cells (Tregs), which function like inhibitors within the immune system, dampening its ability to combat cancer effectively.
A collaborative research effort spearheaded by Mayo Clinic, recently detailed in Cell Reports Medicine, suggests that integrating an advanced next-generation immunotherapy with conventional hormone therapy prior to surgical intervention could address a persistent obstacle in managing early-stage prostate cancer.
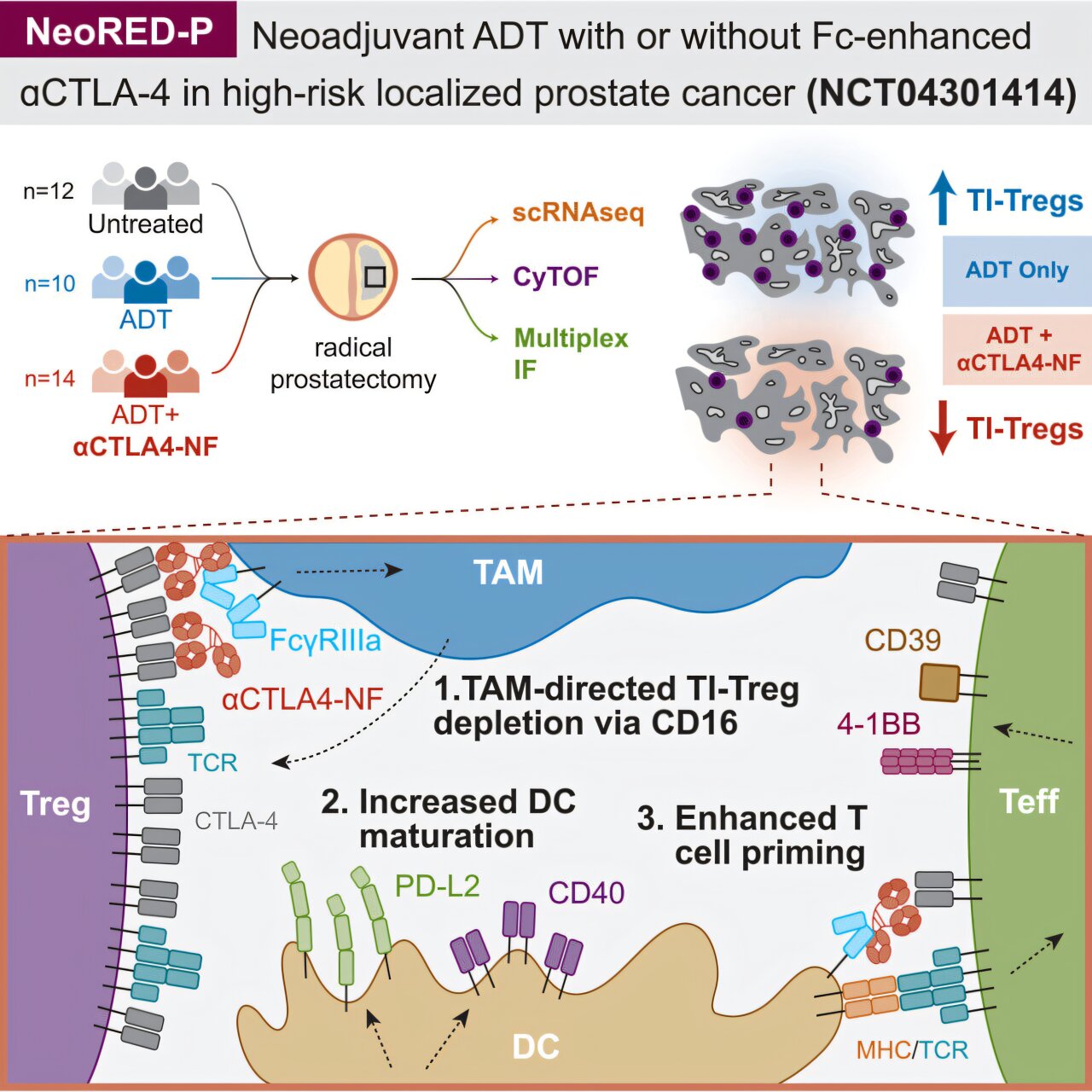
This pioneering early-phase randomized clinical trial marked the first instance where scientists examined whether incorporating a cutting-edge immunotherapy alongside hormone therapy before prostatectomy could mitigate the immunosuppressive effects. Remarkably, the combined regimen successfully lowered Treg levels within prostate tumors. Furthermore, individuals whose tumors exhibited the most substantial Treg reductions demonstrated a higher likelihood of maintaining a cancer-free status throughout the monitoring period post-treatment.
‘This clinical trial offered a distinctive chance to evaluate a novel immunotherapy agent in individuals diagnosed with localized prostate cancer. At this point, they lack metastatic spread, yet they face elevated risks of progression to that advanced stage. For these patients, curative outcomes remain a realistic possibility,’ explained Casey Ager, Ph.D., a researcher specializing in cancer immunology at Mayo Clinic and the study’s lead author.
Androgen deprivation therapy works by depriving prostate cancer cells of essential male hormones such as testosterone, which serve as their primary energy source. While ADT initially triggers beneficial immune responses, these are undermined by Tregs. These cells typically prevent the immune system from excessive reactions against harmless substances or self-tissues. In the context of prostate cancer, as noted by Dr. Ager, Tregs significantly impair the efficacy of immunotherapy interventions.
‘Hormone treatments successfully attract various immune cell types capable of infiltrating, targeting, and destroying tumor cells. Unfortunately, this influx triggers a counter-response where Tregs accumulate, suppressing immune activity and enabling eventual tumor advancement,’ elaborated Dr. Ager.
Dr. Ager, along with fellow Mayo Clinic investigators, partnered with experts from Columbia University Irving Medical Center, Memorial Sloan Kettering Cancer Center, and Bristol Myers Squibb to explore whether targeted Treg suppression could surmount this challenge. Their approach aimed to safely disengage the immune system’s inhibitory mechanisms, thereby enhancing its capacity to combat prostate cancer more robustly.
Designed primarily to assess safety profiles and biological impacts, the study recruited 24 male participants with high-risk localized prostate cancer. Results indicated that supplementing standard hormone therapy with the experimental Fc-enhanced anti-CTLA-4 antibody, BMS-986218, led to a marked decrease in intratumoral Tregs relative to hormone therapy administered in isolation.
‘Achieving selective depletion of Tregs specifically within tumors has long been a primary objective in oncology research. This study allowed us to trial a specially engineered medication optimized for superior Treg elimination compared to prior options. The drug specifically homes in on CTLA-4, a protein abundantly expressed on Tregs, especially those embedded in tumor microenvironments,’ stated Dr. Ager.
These results furnish the inaugural clinical validation that an engineered anti-CTLA-4 treatment can effectively eliminate regulatory T cells directly from within prostate tumors.
Administering the therapy preoperatively enabled researchers to procure and scrutinize substantial portions of the resected prostate glands post-treatment. This was a significant improvement over relying solely on tiny biopsy samples, which typically contain minimal immune cell populations for analysis. Dr. Ager highlighted that this uncommon access facilitated the simultaneous deployment of several state-of-the-art analytical techniques. These methods enabled an extraordinarily detailed mapping of the immunotherapy’s influence on the intricate immune landscape of prostate cancer, extending to the granularity of single immune cells.
Such an in-depth examination of the tumor microenvironment yielded fresh insights into the therapy’s mechanisms of action on immune components, pinpointed which patient subgroups stand to gain the most, and uncovered promising biomarkers that could steer the design of subsequent clinical investigations.
‘Our discoveries affirm the practical viability of deploying immunotherapy in early-stage prostate cancer settings. Moreover, they generate a treasure trove of data essential for crafting and implementing innovative, evidence-driven immunotherapy protocols tailored to these individuals,’ affirmed Dr. Ager.
The research underscores a transformative potential in treating high-risk, early prostate cancers by synergizing advanced immunotherapies with established hormone regimens. This neoadjuvant strategy not only curbs immunosuppressive elements like Tregs but also lays groundwork for personalized medicine approaches. Future studies may expand on these findings, potentially integrating additional agents to further amplify immune activation while minimizing resistance pathways. By addressing the ‘cold’ nature of prostate tumors head-on, this combination therapy opens doors to improved surgical outcomes and prolonged disease-free survival rates. Ongoing research will likely refine dosing schedules, patient selection criteria, and combination partners to maximize therapeutic benefits across diverse prostate cancer profiles.