A recent investigation indicates that at-home testing kits for gut health produce highly variable outcomes and health evaluations, regardless of whether they come from the same manufacturer or different ones. These results, derived from an analysis of kits supplied by seven different providers and detailed in a publication within Communications Biology, emphasize the importance of approaching such test findings with considerable skepticism before making any health-related decisions based on them.
Direct-to-consumer testing kits designed for assessing the gut microbiome offer users detailed insights into the composition of their intestinal bacterial communities. Frequently, these kits go further by providing personalized health evaluations derived from the microbial data. Nevertheless, the reliability and precision of these assessments have long been subjects of debate, as no comprehensive, systematic examination of their effectiveness had been conducted prior to this research effort.
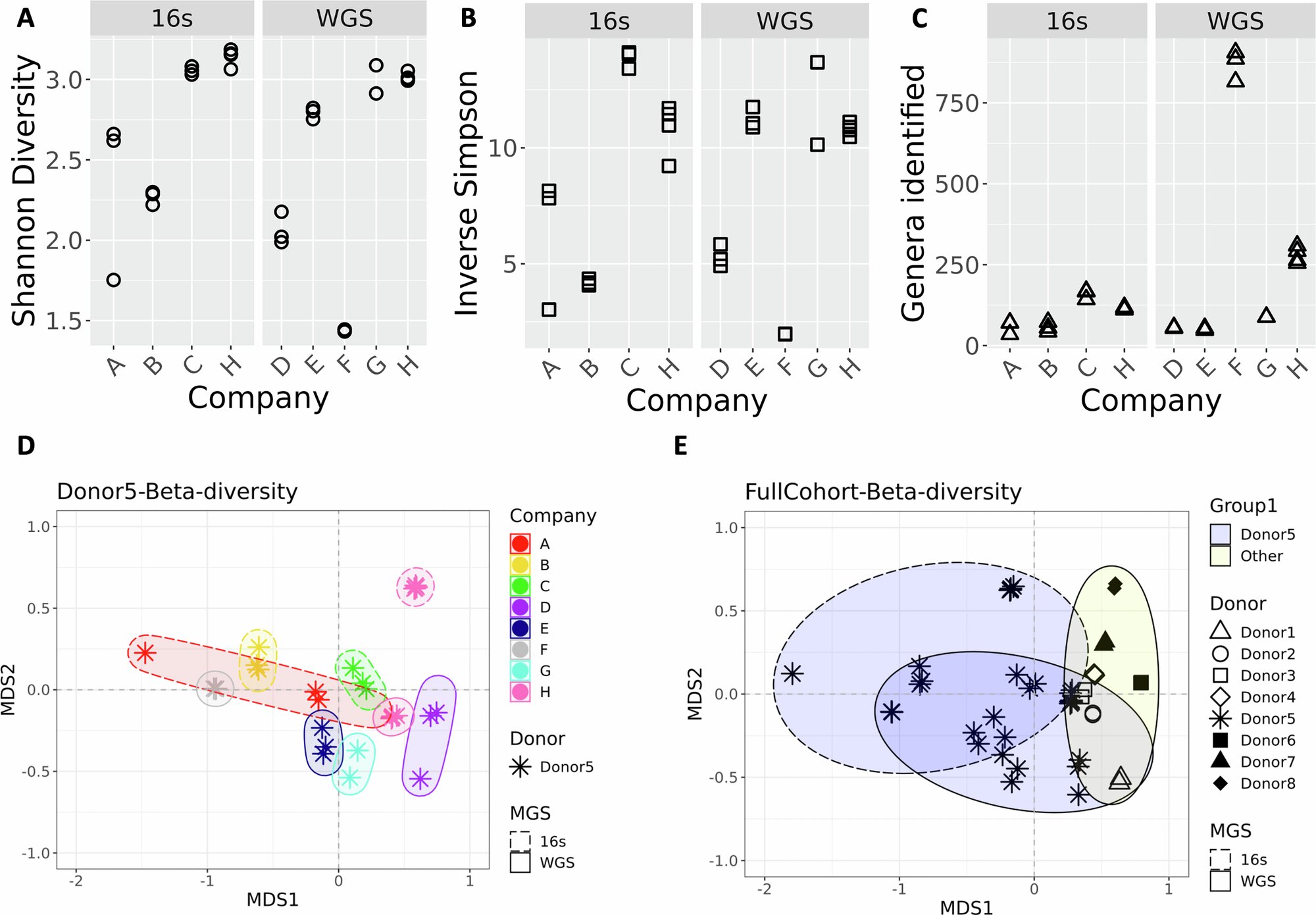
Researchers Stephanie Servetas, Scott Jackson, and their team meticulously evaluated the performance of 21 distinct direct-to-consumer gut microbiome testing kits sourced from seven different companies, which they kept anonymous for the study. Importantly, every single one of these kits was applied to identical composite fecal samples obtained from a single donor. This controlled approach allowed the scientists to isolate variables and pinpoint inconsistencies stemming purely from the testing methodologies employed by each provider.
The study revealed substantial discrepancies in the reported abundances of particular bacterial species within the gut microbiome across the various kits. Notably, the genus Clostridium exhibited some of the most pronounced variations. This genus encompasses several bacterial species that can act as human pathogens, including Clostridioides difficile, which is notorious for causing severe cases of diarrhea and related gastrointestinal infections.
For context, data from the American Gut Project indicated an average abundance of Clostridium at just over 2.5% in typical samples. However, one of the companies participating in this study reported a level five times higher than this benchmark. Even more strikingly, three other companies completely failed to detect the presence of Clostridium in one or more of the analyzed samples, underscoring the erratic nature of these detection capabilities.
Beyond mere bacterial quantification, the researchers also scrutinized the health assessments generated by these kits. In a particularly telling example, when three identical samples were processed using kits from the identical company, the outcomes diverged dramatically: one sample received an “unhealthy” designation, while the remaining two were classified as “healthy.” Such inconsistencies call into question the robustness and reproducibility of the algorithmic interpretations applied to the raw microbial data.
The authors attribute these significant variations to several key factors inherent in the current practices of the direct-to-consumer testing industry. Primarily, there exists a profound absence of standardized procedures for sample collection, handling, processing, and subsequent laboratory analysis. Each company appears to employ its own proprietary protocols, which inevitably introduce variability at multiple stages of the testing pipeline.
Furthermore, the metrics and analytical frameworks utilized by these companies to translate raw microbiome data into actionable health insights differ widely. Without uniform benchmarks or validation against gold-standard laboratory methods, it becomes challenging to ensure that results are comparable or clinically meaningful across providers.
To address these shortcomings, the research team advocates for the development and widespread adoption of standardized sampling protocols, rigorous sample processing guidelines, and harmonized testing methodologies throughout the industry. Implementing such standards could substantially enhance the accuracy, reliability, and trustworthiness of direct-to-consumer gut microbiome testing services, ultimately benefiting consumers who rely on these tools for health monitoring and guidance.
This call for standardization aligns with broader discussions in the field of microbiome research, where reproducibility and methodological consistency are paramount for advancing scientific knowledge and translating it into practical applications. As the market for at-home health tests continues to expand rapidly, regulatory oversight and industry-wide best practices may become essential to protect public health.
The study’s findings serve as a cautionary note for individuals considering these kits. While they offer convenient access to personalized microbiome information, the potential for misleading or contradictory results suggests that users should consult healthcare professionals before acting on the provided recommendations, such as dietary changes or probiotic supplementation.
In summary, this comprehensive evaluation of 21 kits from seven providers using identical samples highlights systemic issues in direct-to-consumer gut health testing. The observed discrepancies in bacterial abundance reporting—particularly for clinically relevant genera like Clostridium—and the inconsistent health categorizations underscore the urgent need for improved standardization. By establishing common protocols for sampling, processing, analysis, and interpretation, the industry can move toward more dependable and useful consumer-facing microbiome assessments.
Publication Details
Stephanie L. Servetas et al, Evaluating the analytical performance of direct-to-consumer gut microbiome testing services, Communications Biology (2026). DOI: 10.1038/s42003-025-09301-3
Journal Reference
Communications Biology
Key Concepts
- Intestinal Flora
- Clostridioides difficile
Relevant Medical Fields
- Gastroenterology
- Common Illnesses and Prevention
This research was conducted by scientists from the National Institute of Standards and Technology, underscoring their commitment to advancing measurement science in biological and health-related domains.