Results from Landmark Randomized Trial on Meningococcal B Vaccine and Gonorrhea Prevention
In a striking departure from what observational research had previously indicated, the meningococcal B vaccine, known as 4CMenB, demonstrates no protective benefit against acquiring gonorrhea. This conclusion emerges from the most extensive randomized controlled trial (RCT) ever performed to evaluate its potential efficacy in this context. The investigation was spearheaded by Griffith University’s Institute for Biomedicine and Glycomics, in collaboration with the Kirby Institute at UNSW Sydney.
Professor Kate Seib from Griffith University unveiled these findings at the Conference on Retroviruses and Opportunistic Infections held in Denver, Colorado. This event brought together leading experts to discuss critical advancements in infectious disease management.
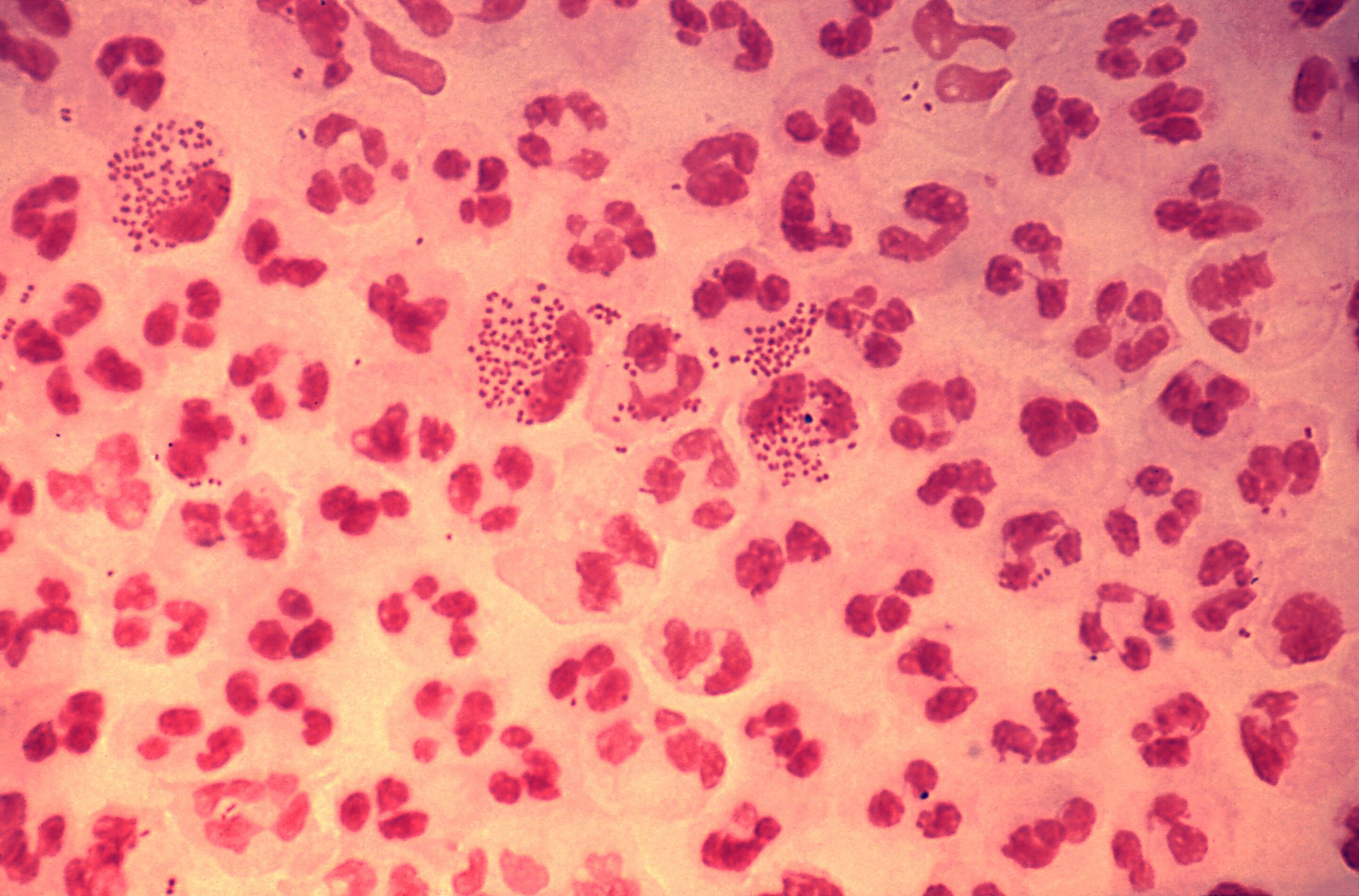
Gonorrhea poses a significant threat to public health worldwide, with growing alarm over the emergence of strains that resist antibiotic treatments. This study specifically targeted gay and bisexual men, a demographic experiencing disproportionately high rates of gonorrhea infections, making them an ideal group for assessing intervention strategies.
The trial adopted a rigorous double-blind, randomized, placebo-controlled design, enrolling 587 gay and bisexual men. Participants were assigned to receive either the meningococcal B vaccine or a saline placebo injection, ensuring unbiased evaluation of the vaccine’s impact.
Professor Kate Seib, the principal investigator from Griffith University, explained, “In both the vaccine and placebo groups, the annual incidence rate of gonorrhea hovered around 48%, underscoring with absolute clarity that the vaccine exerted no influence on preventing new infections.”
She further noted that these outcomes align seamlessly with data from the smaller DOXYVAC open-label trial, which similarly reported a lack of efficacy. “Combined, these robust investigations furnish compelling evidence that the 4CMenB meningococcal vaccine fails to safeguard gay and bisexual men—who face elevated risks—from gonorrhea,” Professor Seib emphasized.
Urgent Need for Alternative Strategies to Combat Gonorrhea
Professor Andrew Grulich from the Kirby Institute, a key leader in the study, expressed profound disappointment: “A vaccine capable of curbing gonorrhea incidence would revolutionize our prevention paradigms. Regrettably, our comprehensive research confirms that 4CMenB does not deliver this protection for gay and bisexual men.”
Approved for use in Australia since 2013, the meningococcal B vaccine remains a highly safe and potent tool for averting meningococcal disease. Professor Grulich reassured participants, stating, “Individuals who opted for the 4CMenB vaccine anticipating gonorrhea protection should rest assured of its safety and its efficacy against certain meningococcal strains. Nevertheless, they must pursue alternative preventive measures, including consistent condom use and routine screening. Accessible services such as sexual health clinics and community testing programs form the bedrock of effective STI prevention.”
This particular RCT focused on gay and bisexual men with documented recent histories of gonorrhea or syphilis, populations predisposed to higher infection risks during the study period. Such selection criteria heightened the likelihood of observing gonorrhea cases, thereby enhancing the trial’s statistical power.
One consideration is whether the participants’ prior exposure to gonorrhea might have diminished any hypothetical vaccine benefits, potentially due to immune preconditioning or other biological factors. While intriguing, this remains speculative.
Although these results may not extrapolate directly to broader populations, they carry profound implications for high-risk gay and bisexual men, guiding clinical recommendations and public health policies tailored to this group.
Brent Mackie, Director of Policy, Strategy, and Research at ACON and a study collaborator, highlighted the value of such research: “Initiatives like this are indispensable, offering the precision required to optimize STI prevention efforts within our communities. We extend our deepest gratitude to all participants—your involvement fortifies sexual health outcomes for everyone.”
Understanding Discrepancies Between Observational Data and RCT Evidence
The GoGoVax RCT, a joint endeavor by Griffith University and the Kirby Institute, was launched in response to multiple observational studies hinting at a correlation between meningococcal B vaccination and lower gonorrhea rates. These preliminary observations prompted several nations to proactively offer the vaccine to high-risk gay and bisexual men as a precautionary measure.
Yet, observational studies are prone to biases introduced by confounding variables—unseen factors that skew associations. For instance, case-control methodologies retrospectively compare disease-affected individuals against unaffected controls to infer causation or prevention pathways.
Professor Grulich elaborated, “Certain case-control analyses juxtaposed meningococcal B vaccination records among gonorrhea patients versus those with chlamydia. They observed unexpectedly low vaccination rates in the gonorrhea cohort, implying a protective role. However, alternative confounders could account for these vaccination disparities between groups, muddying causal interpretations.”
Randomized controlled trials stand as the pinnacle of evidentiary rigor in medical science. Through random assignment, RCTs systematically eliminate confounding influences, isolating true intervention effects.
Professor Grulich affirmed the trial’s integrity: “GoGoVax achieved exemplary randomization, with vaccine and placebo cohorts exhibiting equilibrated confounding variables. This methodological excellence yields the most authoritative proof of causal links.”
Despite the setback, he praised participant dedication: “This outcome, though disheartening, merits recognition of the commitment shown by gay and bisexual men in this research and their enduring fortitude. Collaborative efforts must persist to explore viable gonorrhea prevention avenues.”
Implications for Public Health and Future Research Directions
The trial’s revelations underscore the irreplaceable role of high-quality RCTs in validating therapeutic claims, particularly amid escalating antibiotic resistance in gonorrhea pathogens. With incidence rates climbing and treatment options dwindling, innovative prevention tools remain urgently needed.
Health authorities must recalibrate guidance, emphasizing proven modalities like barrier methods, frequent testing, and partner notification while deprioritizing 4CMenB for gonorrhea control in targeted populations. Concurrently, the vaccine’s established merits against meningococcal disease warrant continued promotion.
This study not only refutes prior hypotheses but also paves the way for next-generation vaccine development, potentially leveraging cross-reactive antigens or novel adjuvants tailored to Neisseria gonorrhoeae’s evasive tactics.
Community engagement, as demonstrated here, proves pivotal in advancing sexual health research. By participating, individuals contribute to evidence-based strategies that safeguard vulnerable groups and curb STI epidemics.
In summary, while the meningococcal B vaccine falls short against gonorrhea, its evaluation illuminates critical pathways forward, reinforcing the necessity for multifaceted, evidence-driven interventions in the ongoing battle against this resilient pathogen.